
(a)
Interpretation:
The name of the given compound has to be given.
Concept introduction:
According to
The order of priority is,
Depending on the number of carbon side chain of the amide, different types of amides can form.
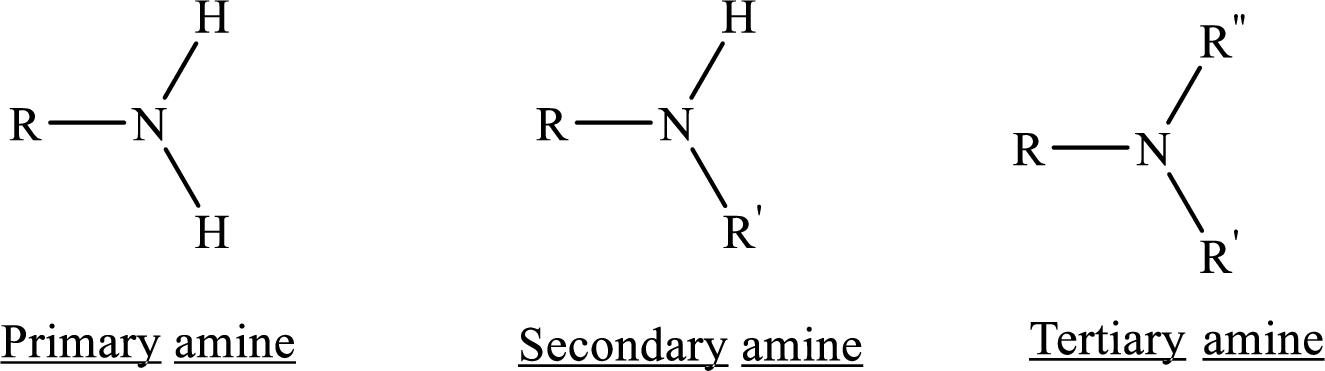
From the name of the compound its structure can be determined.
Primary amines can be named in the IUPAC system in several ways,
For simple amines the suffix – amine is added to the name of the alkyl substituent.
The suffix-amine can be used in place of the final –e in the name of the parent compound.
For a secondary amine an N prefixes the compound giving the shorter carbon chain and its chain prefix name.
For a tertiary amine an N, N prefixes the compound giving the two shorter carbon chains and their side chain prefix names.
Carboxylic acid: One
The IUPAC name for the carboxylic acid is written by replacing the ‘e’ of
If the compound contains amine and a functional group that has higher precedence than the amine group, then the amine group should be indicated with the prefix “amino”
R and S nomenclature: it is used to assign the molecule using CIP rules.
According to Cahn-Ingold-Prelog system,
The group attached to asymmetric center should be ranked based on the
Check the direction of arrow drawn in the direction of decreasing priority. If the arrow points clockwise direction, then the atom has R configuration. If the arrow points counterclockwise direction, then the atom has S configuration. If the group with lowest priority is not bonded by a hatched wedge, then interchange this group (lowest priority) by group bonded to hatched wedge and draw the arrow in priority order but the configuration is assigned as just reverse.
(b)
Interpretation:
The name of the given compound has to be given.
Concept introduction:
According to IUPAC nomenclature, the naming of compound is determined by the priority of the functional group if more than one functional group is present. The carbon attached to the functional group having most priority should get the least number while naming the compound.
The order of priority is,
Amines are the derivatives of ammonia
Depending on the number of carbon side chain of the amide, different types of amides can form.
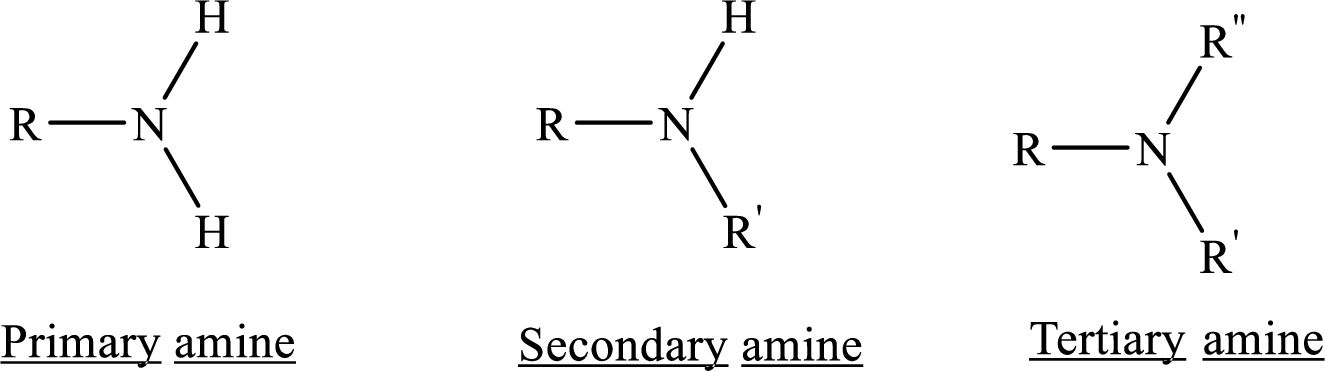
From the name of the compound its structure can be determined.
Primary amines can be named in the IUPAC system in several ways,
For simple amines the suffix – amine is added to the name of the alkyl substituent.
The suffix-amine can be used in place of the final –e in the name of the parent compound.
For a secondary amine an N prefixes the compound giving the shorter carbon chain and its chain prefix name.
For a tertiary amine an N, N prefixes the compound giving the two shorter carbon chains and their side chain prefix names.
Carboxylic acid: One
The IUPAC name for the carboxylic acid is written by replacing the ‘e’ of alkane to ‘oic acid’.
If the compound contains amine and a functional group that has higher precedence than the amine group, then the amine group should be indicated with the prefix “amino”
(c)
Interpretation:
The name of the given compound has to be given.
Concept introduction:
According to IUPAC nomenclature, the naming of compound is determined by the priority of the functional group if more than one functional group is present. The carbon attached to the functional group having most priority should get the least number while naming the compound.
The order of priority is,
Amines are the derivatives of ammonia
Depending on the number of carbon side chain of the amide, different types of amides can form.
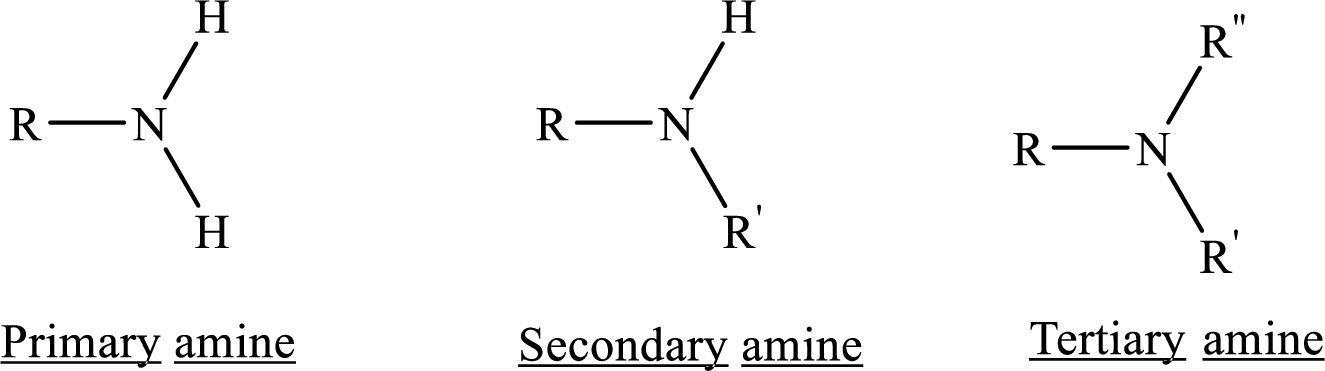
From the name of the compound its structure can be determined.
Primary amines can be named in the IUPAC system in several ways,
For simple amines the suffix – amine is added to the name of the alkyl substituent.
The suffix-amine can be used in place of the final –e in the name of the parent compound.
For a secondary amine an N prefixes the compound giving the shorter carbon chain and its chain prefix name.
For a tertiary amine an N, N prefixes the compound giving the two shorter carbon chains and their side chain prefix names.
Carboxylic acid: One
The IUPAC name for the carboxylic acid is written by replacing the ‘e’ of alkane to ‘oic acid’.
If the compound contains amine and a functional group that has higher precedence than the amine group, then the amine group should be indicated with the prefix “amino”
Common name of amine:
Alkyl groups attached to nitrogen atom of amine group must follow alphabetical order with the suffix “-amine”.
Trending nowThis is a popular solution!

Chapter 23 Solutions
Organic Chemistry
- 1. What are examples of amine compounds that are commonly found in nature and discuss their functions and properties briefly. 2. Show the structure of your example and give its IUPAC name. 3. Describe the reactions that amines may undergo.arrow_forwardDraw the structure for amine. isopropylaminearrow_forwardWrite structural formulas for these amines. Q.) 2-Methyl-1-propanaminearrow_forward
- Describe the Amines compound?arrow_forwardfor the following structures a.) please write the chemical formula for each b.) label which class of compound the structures fall under - using the following: primary amine, secondary amine, tertiary amine, amide, aromatic, alcohol, ester, and carboxylic acidarrow_forwardDraw structural formulas for Q.) The three secondary (2°) amines with the molecular formula C4H11Narrow_forward
 Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning
Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning Macroscale and Microscale Organic ExperimentsChemistryISBN:9781305577190Author:Kenneth L. Williamson, Katherine M. MastersPublisher:Brooks Cole
Macroscale and Microscale Organic ExperimentsChemistryISBN:9781305577190Author:Kenneth L. Williamson, Katherine M. MastersPublisher:Brooks Cole

