
Concept explainers
Interpretation:
In the nitration of phenol (
Concept introduction:
The substituent on the benzene ring prior to substitution dictates the regiochemistry of the reaction. Some substituents are designated as ortho/para directors because they lead to product mixtures consisting primarily of the ortho- and para-disubstituted products. Other substituents are designated as meta directors because they favor the formation of the meta-disubstituted product. The results from nitration reactions are summarized in Table 23-1:
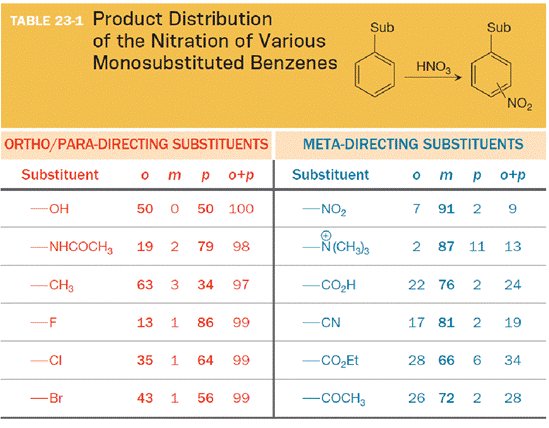
In the nitration of phenol (
Want to see the full answer?
Check out a sample textbook solution
Chapter 23 Solutions
ORG CHEM W/ EBOOK & SW5 + STUDY GUIDE
- Draw the mechanism for this reactionarrow_forwardThe reaction shown here proceeds via a carbocation rearrangement. Draw a complete, detailed mechanism to account for the product. Explain why the carbocation rearrangement is favorable. CH;OH Brarrow_forwardts] Draw the mechanism for the reaction listed below. acetone benzaldehyde NaOHarrow_forward
- Draw the mechanism for this reaction. HO H,SO4.arrow_forwardConsider the intramolecular nucleophilic substitution reaction shown here. Does the stereochemistry of the product suggest an Sn1 or SŅ2 mechanism? Draw the complete mechanism for this reaction, including curved Br OH NaOH CH3 H3C CH3 Br H3C H arrows.arrow_forwardIf the anionic species shown were to eliminate a leaving group, the three possi- bilities would be H3C¯, CI, or CH30¯. Draw the curved arrow notation and the products for each of these elimination steps. Which is the major product? Why? CIarrow_forward
- Shown below is a two-step mechanism beginning with nucleophilic attack of water, and subsequent deprotonation with a base. Draw the arrows for the mechanism for both step 1 and step 2 and draw the intermediate product of in the box. HH H-O OH H₂O Barrow_forwardIf H* is eliminated from the carbocation shown here in an electrophile elimination step, then three possible constitutional isomers can form. Draw the mechanism for the formation of all three of those products. H2O + ?arrow_forwardCompare the following reactions. Which is faster? Using a Free Energy Diagram, justify your choice. Br NaOH Br NaOHarrow_forward
 Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
