
(a)
Interpretation:
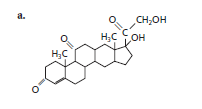
Whether the following structure is classified as fatty acid, triglyceride, phospholipid, sterioid or wax should be determined.
Concept introduction:
The molecules which contains hydrocarbons and not soluble in water but soluble in nonpolar solvents is known as lipid.
Lipids are classified as:
Triglycerides, Phospholipids and, Steroids and Waxes.
The building blocks of fat in our bodies and in food are known as fatty acids. Fatty acid consists of carboxylic acid with long aliphatic chain which can be saturated or unsaturated.
The given structure is classified as steroid as it contains multiple cyclic rings.
Fatty acid consists of carboxylic acid with long aliphatic chain which can be saturated or unsaturated. Generally, fatty acids exist as: phospholipids, triglycerides and cholesteryl esters. These are important structural components for cells.
Fats and oils consist of molecules known as triglycerides (contain ester bonds).
The given structure is:
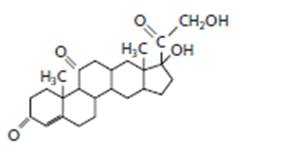
The skeleton of given structure is similar to steroid as it contains many cyclic structures. General structure of steroid contains 17 carbon atoms which are linked with each other in four fused rings, three rings contains six carbon atoms that is cyclohexane rings and one is five membered ring that is cyclopentane.
Thus, in the given structure, four cyclohexane rings are fused and one five membered ring that is cyclopentane is present along with a
Hence, the given structure is classified as steroid.
(b)
Interpretation:
Whether the following structure is classified as fatty acid, triglyceride, phospholipid, sterioid or wax should be determined.
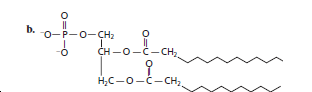
Concept introduction:
The molecules which contains hydrocarbons and not soluble in water but soluble in nonpolar solvents is known as lipid.
Lipids are classified as:
Triglycerides, Phospholipids and, Steroids and Waxes
The building blocks of fat in our bodies and in food are known as fatty acids. Fatty acid consists of carboxylic acid with long aliphatic chain which can be saturated or unsaturated.
The given structure is classified as phospholipid as it contains a phosphate group, diglyceride and two straight chains of carbon atoms.
Fatty acid consists of carboxylic acid with long aliphatic chain which can be saturated or unsaturated. Generally, fatty acids exist as: phospholipids, triglycerides and cholesteryl esters. These are important structural components for cells.
Fats and oils consist of molecules known as triglycerides (contain ester bonds).
The given structure is:
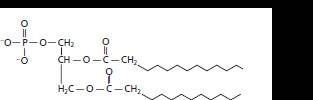
The skeleton of given structure is similar to phospholipids. General structure of phospholipids contains a glycerol molecule(s), phosphate group and two fatty acids.
Thus, in the given structure, two fatty acids are present that are two straight chains of carbon atoms along with a phosphate group and two glyceride group that is diglyceride.
Hence, the given structure is classified as phospholipid.
Chapter 23 Solutions
Chemistry: Matter and Change
Additional Science Textbook Solutions
Chemistry: Structure and Properties (2nd Edition)
Chemistry: The Central Science (14th Edition)
CHEMISTRY-TEXT
Organic Chemistry (9th Edition)
Chemistry: The Central Science (13th Edition)
General Chemistry: Principles and Modern Applications (11th Edition)
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY





