
Concept explainers
Interpretation:
The cycloaddition reaction involving two molecules of cyclopentene is as follows:
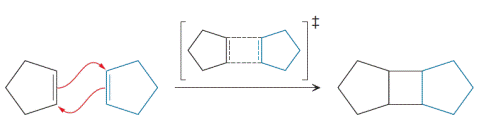
Whether the transition state is
Concept introduction:
The Diels-Alder reaction is a single step concerted reaction, whereby all of the bonds are formed and broken simultaneously. Thus, the reaction proceeds through a cyclic transition state, which makes it a pericyclic reaction. All Diels-Alder reactions involve the cyclic flow of six π electrons. The number of electrons is important because these electrons are delocalized over a complete ring in the transition state. Six electrons is a Hückel number of electrons (i.e., an odd number of pairs), and the species is aromatic when the electrons are delocalized over an entire ring. Thus, Diels-Alder reaction proceeds through an aromatic transition state.
Want to see the full answer?
Check out a sample textbook solution
Chapter 24 Solutions
ORGANIC CHEMISTRY E-BOOK W/SMARTWORK5
- This is a hydrogenation and carbene addition problem. How do I find what the missing reactant is of the reaction?arrow_forwardIdentify the mode of ring closure for the given electrocyclic reaction. Also, identify if the the indicated hydrogens are cis or trans.arrow_forwardDefine Cycloaddition Reactions ?arrow_forward
- Why does catalytic hydrogenation of alkenes proceed through syn addition? Provide a detailed explanation and illustrate as needed (5 sentences only).arrow_forwardThis is a hydrogenation and carbene addition problem. How do I draw the structure of the major organic product of the reaction?arrow_forwardImagine the following substitution reaction of A+B -> C withthe effect of concentration on rate as shown in the table. Identify the mechanism as SN1 or SN2 and write the rate law equation.arrow_forward
- What is/are the monochlorination product(s) of each of the following reactions? Disregard stereoisomers.arrow_forwardIdentify the mode of ring closure for each of the following electrocyclic reactions. a. Are the indicated hydrogens cis or transarrow_forwardIdentify the pericyclic reactions in the following reaction schemes. Give the complete reaction name and indicate the course of the reaction with the aid of the arrow notation.arrow_forward
- Explain difference of reactions. Why addition of peroxides reverses regioselectivity of HBr addition to alkenes by showing mechanism, intermediate stability.arrow_forwardThis is a hydrogenation and carbene addition problem. How do I draw the structure of the major organic product of the reaction shown?arrow_forwardDetermine the products of the following reactions, no reaction mechanism is necessary. In part b), where the question mark is, conditions can be proposed for any product of interest or that is feasible to obtain with the compound of the previous reaction.arrow_forward

