
Organic Chemistry (9th Edition)
9th Edition
ISBN: 9780321971371
Author: Leroy G. Wade, Jan W. Simek
Publisher: PEARSON
expand_more
expand_more
format_list_bulleted
Concept explainers
Textbook Question
Chapter 24, Problem 24.40SP
Lipoic acid is often found near the active sites of enzymes, usually bound to the peptide by a long flexible amide linkage with a lysine residue.
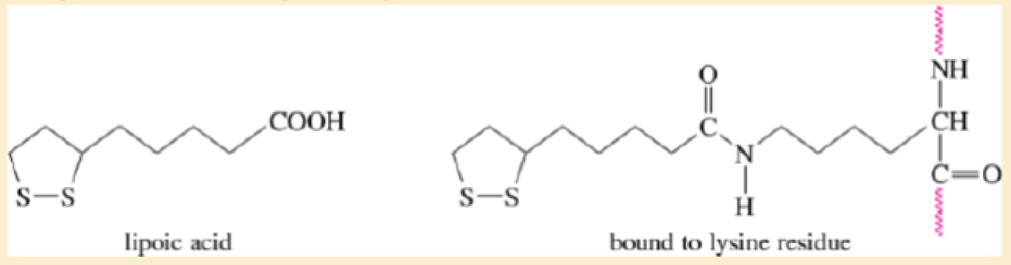
- a. Is lipoic acid a mild oxidizing agent or a mild reducing agent? Draw it in both its oxidized and reduced forms.
- b. Show how lipoic acid might react with two Cys residues to form a disulfide bridge.
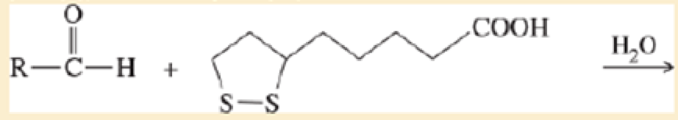
- c. Give a balanced equation for the hypothetical oxidation or reduction, as you predicted in part (a), of an
aldehyde by lipoic acid.
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
A tripeptide undergoes complete hydrolysis and the resulting mixture contains only phenylalanine and glycine. Draw all possible sequences for the original tripeptide.
Originally the amino acid itself was considered to be used as a flavor enhancer, but MSG was determined to have better water solubility. Why is that the case?
1. (a) Draw a circle around one amino acidin the structure. (
b) Draw a box around one amino acid side chain.
(c) Indicate whether the side chain you chose is hydrophobic or hydrophilic.
(d) Draw an arrow pointing to a peptide bond
2. If the peptide segment shown above was part of a beta sheet, which of its functional groups would be involved in forming and stabilizing the beta sheet?
3. Which amino acid (also called amino acid residue)would be more likely to occur on the surface of a protein: an aspartate residue or a methionine residue? Explain your choice.
Chapter 24 Solutions
Organic Chemistry (9th Edition)
Ch. 24.2A - Draw three-dimensional representations of the...Ch. 24.2A - Prob. 24.2PCh. 24.2B - The herbicide glyphosate (Roundup) kills plants by...Ch. 24.4 - Draw the structure of the predominant form of a....Ch. 24.4 - Draw the resonance forms of a protonated guanidino...Ch. 24.4 - Although tryptophan contains a heterocyclic amine,...Ch. 24.4 - Prob. 24.7PCh. 24.4 - Prob. 24.8PCh. 24.5A - Show how the following amino acids might be formed...Ch. 24.5B - Prob. 24.10P
Ch. 24.5C - Prob. 24.11PCh. 24.5C - Show how you would use a Strecker synthesis to...Ch. 24.6 - Suggest how you would separate the free i-ammo...Ch. 24.7A - Propose a mechanism for the acid-catalyzed...Ch. 24.7A - Give equations for the formation and...Ch. 24.7B - Prob. 24.16PCh. 24.7C - Prob. 24.17PCh. 24.8B - Draw the complete structures of the following...Ch. 24.9C - Prob. 24.19PCh. 24.9C - Prob. 24.20PCh. 24.9C - Prob. 24.21PCh. 24.9E - Prob. 24.22PCh. 24.9E - Prob. 24.23PCh. 24.10A - Propose a mechanism for the coupling of acetic...Ch. 24.10B - Show how you would synthesize Leu-Gly-Ala-Val-Phe...Ch. 24.10B - Show how solid-phase peptide synthesis would be...Ch. 24 - a. The isoelectric point (pl) of phenylalanine is...Ch. 24 - Prob. 24.28SPCh. 24 - Prob. 24.29SPCh. 24 - Prob. 24.30SPCh. 24 - Prob. 24.31SPCh. 24 - Suggest a method for the synthesis of the...Ch. 24 - Prob. 24.33SPCh. 24 - Write the complete structures for the following...Ch. 24 - The following structure is drawn in an...Ch. 24 - Prob. 24.36SPCh. 24 - Prob. 24.37SPCh. 24 - Show the steps and intermediates in the synthesis...Ch. 24 - Prob. 24.39SPCh. 24 - Lipoic acid is often found near the active sites...Ch. 24 - Prob. 24.41SPCh. 24 - Prob. 24.42SPCh. 24 - Prob. 24.43SPCh. 24 - Complete hydrolysis of an unknown basic...Ch. 24 - Prob. 24.45SPCh. 24 - Prob. 24.46SPCh. 24 - Prob. 24.47SP
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- According to the paper, F486 is a phenylalanine residue (i.e., an amino acid that is part of a chain that forms a protein) on SARS-CoV-2. It has an important interaction with ACE2 (best seen in Fig. 1(C) and Fig. 2(A)). Looking at this phenylalanine and the green residues surrounding it, which statement best describes the interaction that could be occurring? a) It forms a hydrogen bond with Y83. b) It forms a dipole-dipole interaction with M82. c) It forms London dispersion forces with surrounding residues. d) It encounters repulsions from surrounding residues.arrow_forwardA tripeptide on hydrolysis produced glycine, alanine and leucine. The structures of these amino acids are shown below. On reaction with Edman’s reagent, leucine was released as the phenylhydantoin. Treatment of the tripeptide with carboxypeptidase gave glycine. Draw the structure of the tripeptide.arrow_forwarda) A solution of amino acid having carboxylic side chain was titrated against NaOH. If initial pH of the solution was 2.8. Describe the state of the functional groups of the amino acid at the initial pH and at 10.5.b) Briefly explain how you will separate amino acids mixture using the charge difference between the amino acids. c) A given glycine solution was titrated against NaOH and pH of the final solution was 4.8. Given that pKa1 and pKa2 of glycine are 2.34 and 9.60, respectively. Calculate the concentration of the dissociated amino acid in terms of the undissociated amino acid, if [x] and [y] are the concentrations of dissociated and undissociated amino acid, respectively.arrow_forward
- Another method to form a peptide bond involves a two-step process:[1] Conversion of a Boc-protected amino acid to a p-nitrophenyl ester.[2] Reaction of the p-nitrophenyl ester with an amino acid ester.a. Why does a p-nitrophenyl ester “activate” the carboxy group of the first amino acid to amide formation? b. Would a p-methoxyphenyl ester perform the same function? Why or why not?arrow_forwardDraw Tryptophan in a peptide bond. Explain all of the bonds using valence bond theory (VBT). Next, explain where VBT fails and how molecular orbital theory is a better description of key regions of this amino acid and the peptide bonds it forms with other residuesarrow_forwardindicate the RIGHT alternative: (a) The Zwitterion form of an amino acid exists only at a point pH value. (b) In a peptide bond there is free rotation at the C-N bond. (c) In a polypeptide, the terminal carboxyl group may be present in its amide form. (d) At a pH greater than pI, an amino acid tends to move towards the cathode in an electrophoresis. (e) At any pH below pI, the predominant form of an amino acid is negatively charged.arrow_forward
- The amino acid (S)-alanine has the physical characteristics listed under the structure. a.What is the melting point of (R)-alanine? b.How does the melting point of a racemic mixture of (R)- and (S)-alanine compare to the melting point of (S)-alanine? c. What is the specific rotation of (R)-alanine, recorded under the same conditions as the reported rotation of (S)-alanine? d.What is the optical rotation of a racemic mixture of (R)- and (S)-alanine? e.Label each of the following as optically active or inactive: a solution of pure (S)alanine; an equal mixture of (R)- and (S)-alanine; a solution that contains 75% (S)- and 25% (R)-alanine.arrow_forward. True/ False A. Amino acids found in the living systems are generally in the D-form B. The R-group attached to alfa carbon atom of amino acids are unique for each amino acids. C. Of the 20 essential amino acids, glycine is the only amino acid which is achiral. D. Ions of heavy metals Hg2+ or Pb2+ denature proteins by combining with the amino groups. E. Denaturation of protein involves the breakdown of secondary, tertiary and quaternary structure. F. Cysteine is the only amino acid that can form a disulfide bridge. G. Both the poly saccharides and proteins are polymers. H. Hydrogen bonding is responsible for the secondary structure of Proteins. I. When proteins are hydrolyzed, smaller peptides and amino acids are produced. J. When simple lipids are hydrolyzed glycerol an alcohol as well as fatty acids and their salts are produced.arrow_forwardWrite out the steps needed to synthesize the following peptide using the Merrifield method.arrow_forward
- Another method to form a peptide bond involves a two-step process Reaction of the p-nitrophenyl ester with an amino acid ester. Why does a p-nitrophenyl ester “activate” the carboxy group of thefirst amino acid to amide formation?arrow_forwardConsider the following tetrapeptide, written in a sequence according to normal convention, from left to right: Residue 1 is acidic; Residue 2 is cysteine (an amino acid containing a –CH2SH R-group); Residue 3 is proline (a large hydrophobic amino acid); Residue 4 is basic. Of the following four general statements, which one is TRUE regarding this tetrapeptide? This tetrapeptide does not have the ability to form a disulfide linkage with an identical tetrapeptide. Residue 2 belongs to the amino acid category known as "basic." 3 of these 4 responses are correct This tetrapeptide could be part of an alpha-helix, since the large size of the proline would not have any impact on the consistency of the spiral shape. The acidic residue (Residue #1) is at the amino terminus of this tetrapeptide.arrow_forwardBy using only amide/Peptide bond forming rxn and a lKylation rxns. Provide all the reaction steps with the reagent needed and conditions with out showing the mechanism for each step to form the product. Note: Use protecting groups like (Boc, Fmoc, and others) H2NNH2+arrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 Organic And Biological ChemistryChemistryISBN:9781305081079Author:STOKER, H. Stephen (howard Stephen)Publisher:Cengage Learning,
Organic And Biological ChemistryChemistryISBN:9781305081079Author:STOKER, H. Stephen (howard Stephen)Publisher:Cengage Learning, General, Organic, and Biological ChemistryChemistryISBN:9781285853918Author:H. Stephen StokerPublisher:Cengage Learning
General, Organic, and Biological ChemistryChemistryISBN:9781285853918Author:H. Stephen StokerPublisher:Cengage Learning
 EBK A SMALL SCALE APPROACH TO ORGANIC LChemistryISBN:9781305446021Author:LampmanPublisher:CENGAGE LEARNING - CONSIGNMENT
EBK A SMALL SCALE APPROACH TO ORGANIC LChemistryISBN:9781305446021Author:LampmanPublisher:CENGAGE LEARNING - CONSIGNMENT

Organic And Biological Chemistry
Chemistry
ISBN:9781305081079
Author:STOKER, H. Stephen (howard Stephen)
Publisher:Cengage Learning,

General, Organic, and Biological Chemistry
Chemistry
ISBN:9781285853918
Author:H. Stephen Stoker
Publisher:Cengage Learning


EBK A SMALL SCALE APPROACH TO ORGANIC L
Chemistry
ISBN:9781305446021
Author:Lampman
Publisher:CENGAGE LEARNING - CONSIGNMENT
Biomolecules - Protein - Amino acids; Author: Tutorials Point (India) Ltd.;https://www.youtube.com/watch?v=ySNVPDHJ0ek;License: Standard YouTube License, CC-BY