
(a)
Interpretation:
The resolution for species B and C from the given data should be determined.
Concept introduction:
The resolution of the column is defined as the separation of two species of the column.
Answer to Problem 26.16QAP
The resolution is
Explanation of Solution
Given:
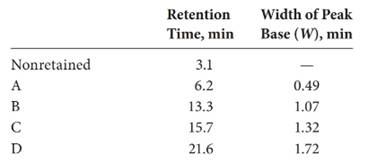
The expression of resolution of the column is:
Here, the retention time of species
Substitute
Thus, the resolution is
(b)
Interpretation:
The selectivity factor for species B and C from the given data should be determined.
Concept introduction:
The resolution of the column is defined as the separation of two species of the column.
Answer to Problem 26.16QAP
The selectivity factor is
Explanation of Solution
The expression of selectivity factor is:
Here, the non-retained retention time is
Substitute
Thus, the selectivity factor is
(c)
Interpretation:
The length of column necessary to separate B and C species with a resolution of
Concept introduction:
The resolution of the column is defined as the separation of two species of the column.
Answer to Problem 26.16QAP
The length of column necessary to separate the two species with a resolution of
Explanation of Solution
The expression of length of the column is:
Here, the number of plates needed to separate the two species is
The expression of relation of the resolution and number of plates is:
Here, the number of plates needed is
Substitute
Substitute
Thus, the length of column necessary to separate the two species with a resolution of
(d)
Interpretation:
The time required to separate B and C species on the column of part c should be determined.
Concept introduction:
The resolution of the column is defined as the separation of two species of the column.
Answer to Problem 26.16QAP
The time required to separate the two species on the column of part c is
Explanation of Solution
The expression of the relation of time required to separate the two species on the column is:
Here, the given resolution is
Substitute
Thus, the time required to separate the two species on the column of part c is
Want to see more full solutions like this?
Chapter 26 Solutions
Principles of Instrumental Analysis
- From the data in Problem 26-14, calculate for species C and D) (a) the resolution. (b) the length of column necessary to separate the two species with a resolution of 2.5.arrow_forwardTwo species A and B are known to have water/hexane partition coefficient of 5.99 and 6.16.They are separated by elution on silica gel with hexane as eluent. The ratio for the packingVS/VM =0.425a. Calculate the retention factor for each soluteb. Calculate the selectivity factorc. How many plates are needed to provide a resolution of 1.5?d. What column length should be used to provide a resolution of 1.5 if the plate height ofthe packing is 1.5 ×10-3cm?e. If the flow rate is 6.75 cm min-1, how long will it take to elute the two species? (the question wanted to be answered)arrow_forwardIn color chromatography of plant pigments, what complications would a dried-out column (solvent level is below the top of the silica) introduce to the elution and isolation of pigments?arrow_forward
- PART A: Substance A was found to have a retention times of 3.15 minutes on a 70.00-cm column. An unretained species passed through the column in 0.50 minutes. The peak width (at base) for A was 0.60 minutes. Using this information, determine the following: 1. What is the retention factor for analyte A? 2. What is the plate count for this column? 3. What is the height equivalent of a theoretical plate? PART B: Match each statement with one of these terms. A Partition coefficient B Mobile phase C Elution D Longitudinal diffusion E Stationary phase F Eddy diffusion G Retention factor H Plate number I Dead time J Retention time K Selectivity factor L Column resolutionarrow_forwardSubstances A and B have retention times of 12.75 and 14.28 min, respectively, on a 30.0-cm column. An unretained species passes through the column in 1.10 min. The peak widths (at base) for A and B are 1.11 and 1.27 min, respectively. What is the resolution?arrow_forwardExplain the purpose of the collision cell in interface between inductively coupled plasma and mass spectrometer.arrow_forward
- In order to improve the peptide separation by using a HPLC system, trifluoroacetic acid acts as mobile-phase modifier was added during the preparation of mobile phase. The preparation was performed by a postgraduate student as following:“2.851 g trifluoroacetic acid (MW: 114.02 g/mol) was made up to 500 cm3 in a graduated flask. To this solution, 50 cm3 of ethanol was added, and after mixing the mobile phase was placed in the solvent reservoir and pumping was commenced at 1.5 cm3 min-1.”Based on the given preparation procedure, identify THREE mistakes that were made.arrow_forwardHow the 2D Thin layer Chromatography separation processes are worked? Please shortly wtite at your own words. Answer should be to the point (5-6 lines maximum).arrow_forwardA method for the analysis of Ca2+ in water suffers from an interference in the presence of Zn2+. When the concentration of Ca2+ is 100 times greater than that of Zn2+, an analysis for Ca2+ gives a relative error of +0.5%. What is the selectivity coefficient for this method?arrow_forward
- Two species A and B are known to have water/hexane partition coefficient of 5.99 and 6.16. They are separated by elution on silica gel with hexane as eluent. The ratio for the packing VS/VM =0.425a) What column length should be used to provide a resolution of 1.5 if the plate height ofthe packing is 1.5 ×10-3 cm? b) If the flow rate is 6.75 cm min-1, how long will it take to elute the two species?arrow_forwardWhat is the role of stationary phase and mobile phase in Chromatography? Please shortly answer at your own words. Answer should be to the point (specific 4-5 lines).arrow_forwardIf satisfactory quantitative data are to be obtained from chromatograms, what parameters must be manipulated and how? O increasing the column length O increasing the internal diameter of the column O increasing the particle size of the column packing O all of the abovearrow_forward
 Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning


