
Concept explainers
(a)
Interpretation:
Corresponding range of wavelength and frequencies should be calculated for given wavenumbers.
Concept introduction:
(b)
Interpretation:
The bonds responsible for the absorptions labeled A, B, C, and D in the two infrared spectrashown here should be identified.
One spectrum is for acetone and the other spectrum is for 1-propanol, both of which are colorless liquids. From the two spectra, the spectra of acetone should be determined.
Concept introduction:
Acetone; - has one C=O bond, 2 C-C bonds, 6 C-H bonds.
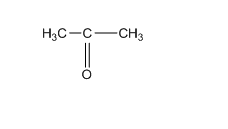
1-propanol; - has one C-O bond, 2 C-C bonds, 7 C-H bonds and one O-H bond.
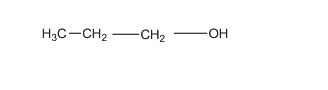
(c)
Interpretation:
The structure of the compound should be determined on the basis of given information:
Want to see the full answer?
Check out a sample textbook solution
Chapter 26 Solutions
General Chemistry: Principles and Modern Applications - With Solutions Manual and Modified MasteringChemistry Code
- TRUE OR FALSE All atoms play a role in the physicochemical and pharmacologic properties of a molecule. Sophistication and advancement in technology are the main driving forces of drug development. Organic medicinal chemistry is the study of matter, specifically, all carbon-containing compounds. A functional group will always have the same physicochemical properties, regardless of its neighboring atoms in a molecular structure. Addition of a single methyl group in a drug molecule may result to changes in physical, chemical, pharmacokinetic, and pharmacologic properties. Steric effects occur when bulky and large functional groups are near each other in a molecular structure. Molecules that are highly capable of hydrogen bonding tend to be more soluble in nonpolar solvents.arrow_forwardChoose three objects in your home that are considered as organic compounds. Write down whether it is an alkane, alkene, alkyne, alcohol, carboxylic acid, ether, aldehyde, ketone, ester, amine. Also include its structure, functional group (group found in homologous series such as hydroxyl [-OH], carboxyl [-COOH], etc.), at least three properties, and other examples of the organic compounds in real life (Note: These other examples must be for each functional group identified).arrow_forwardConsider an organic molecule with the molecular formula C13H10O. a. Propose a chemically correct structure for this molecule that contains a ketone. b. Propose a chemically correct structure for this molecule that contains a functional group other than a ketonearrow_forward
- (Picture attached) Functional Groups: 1. Benzene 2. Halogen 3. Carboxyl 4. Hydroxyl Identify the functional groups that the 2 molecules contain. Note: each functional group can be used more than once. Put in numerical order with no space. Sucarlose = Ibuprofen =arrow_forwardWhat is a hydrocarbon? What is the difference between a saturated hydrocarbon and an unsaturated hydrocarbon? Distinguish between normal and branched hydrocarbons. What is an alkane? What is a cyclic alkane? What are the two general formulas for alkanes? What is the hybridization of carbon atoms in alkanes? What are the bond angles in alkanes? Why are cyclopropane and cyclobutane so reactive? The normal (unbranched) hydrocarbons are often referred to as straight-chain hydrocarbons. What does this name refer to? Does it mean that the carbon atoms in a straight-chain hydrocarbon really have a linear arrangement? Explain. In the shorthand notation for cyclic alkanes, the hydrogens are usually omitted. How do you determine the number of hydrogens bonded to each carbon in a ring structure?arrow_forwardHow many electron pairs are shared when a triple bond exists between two carbon atoms? What must he the geometric arrangement around the carbon atoms in a triple bond? Draw the Lewis structure of a simple molecule that contains a triple bond.arrow_forward
- What are the functional groups in Organic chemistry? Write their structures and give at least three examples in each group.arrow_forwardIs there any difference between 2-methylpropane and isobutane? Why is the structure in the image not named 2-methylpropane but isobutane. What is meant by "iso"?arrow_forwardOne or more ways in which this General Chemistry, Organic Chemistry and Biochemistry course changed the way that you see and understand the world?arrow_forward
- Org. Chemistry - CANNOT be hand-drawn! 2,6-dimethyloct-2-ene Functional Groups illustrate the structural formula of your molecule (expanded or condensed) and indicate the functional groups present by highlighting, circling, or color-coding each and labeling the group. See my attached image - I need help correcting this with a new computerized illustration Thank you!arrow_forwardOrganic Chemistry HW: 2,6-dimethyloct-2-ene cannot be handwritten, explanation of print the structural formula of your molecule (expanded or condensed) and indicate the functional groups present by highlighting, circling, or color-coding each and labeling the group.arrow_forwardWhat is a functional group? a. a group of atoms with a specific purpose or function b. a collection of atoms that work well together c. a group of atoms in an organic molecule that is the site of a chemical reactionarrow_forward
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
 Chemistry: An Atoms First ApproachChemistryISBN:9781305079243Author:Steven S. Zumdahl, Susan A. ZumdahlPublisher:Cengage Learning
Chemistry: An Atoms First ApproachChemistryISBN:9781305079243Author:Steven S. Zumdahl, Susan A. ZumdahlPublisher:Cengage Learning World of Chemistry, 3rd editionChemistryISBN:9781133109655Author:Steven S. Zumdahl, Susan L. Zumdahl, Donald J. DeCostePublisher:Brooks / Cole / Cengage Learning
World of Chemistry, 3rd editionChemistryISBN:9781133109655Author:Steven S. Zumdahl, Susan L. Zumdahl, Donald J. DeCostePublisher:Brooks / Cole / Cengage Learning Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning
Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning Introductory Chemistry: A FoundationChemistryISBN:9781337399425Author:Steven S. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
Introductory Chemistry: A FoundationChemistryISBN:9781337399425Author:Steven S. Zumdahl, Donald J. DeCostePublisher:Cengage Learning





