
Concept explainers
(a)
Interpretation:
Whether the following pair of structures is identical molecules, enantiomers or isomers of some other sort:
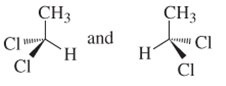
Concept introduction:
Structural or constitutional isomers can be defined as the molecules with same molecular formula and different structural formula. They are molecules in which the bonded atoms have different bonding with each other although the number of molecules is same. Identical molecules are molecules which have same structural formula and same molecular formula.
Enantiomers are optical isomers which can rotate the plane polarized light either clockwise or anticlockwise. These molecules must have at least one chiral C atom which is bonded with four different groups. They are named as R and S configuration. For naming the enantiomer as R or S-enantiomer, the Cahn-Ingold-Prelog rule is followed.
(b)
Interpretation:
Whether the following pair of structures is identical molecules, enantiomers or isomers of some other sort:
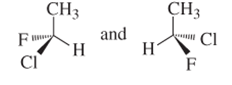
Concept introduction:
Structural or constitutional isomers can be defined as the molecules with same molecular formula and different structural formula. They are molecules in which the bonded atoms have different bonding with each other although the number of molecules is same. Identical molecules are molecules which have same structural formula and same molecular formula.
Enantiomers are optical isomers which can rotate the plane polarized light either clockwise or anticlockwise. These molecules must have at least one chiral C atom which is bonded with four different groups. They are named as R and S configuration. For naming the enantiomer as R or S-enantiomer, the Cahn-Ingold-Prelog rule is followed.
(c)
Interpretation:
Whether the following pair of structures is identical molecules, enantiomers or isomers of some other sort:
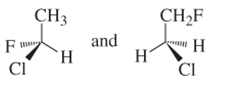
Concept introduction:
Structural or constitutional isomers can be defined as the molecules with same molecular formula and different structural formula. They are molecules in which the bonded atoms have different bonding with each other although the number of molecules is same. Identical molecules are molecules which have same structural formula and same molecular formula.
Enantiomers are optical isomers which can rotate the plane polarized light either clockwise or anticlockwise. These molecules must have at least one chiral C atom which is bonded with four different groups. They are named as R and S configuration. For naming the enantiomer as R or S-enantiomer, the Cahn-Ingold-Prelog rule is followed.
(d)
Interpretation:
Whether the following pair of structures is identical molecules, enantiomers or isomers of some other sort:
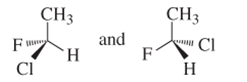
Concept introduction:
Structural or constitutional isomers can be defined as the molecules with same molecular formula and different structural formula. They are molecules in which the bonded atoms have different bonding with each other although the number of molecules is same. Identical molecules are molecules which have same structural formula and same molecular formula.
Enantiomers are optical isomers which can rotate the plane polarized light either clockwise or anticlockwise. These molecules must have at least one chiral C atom which is bonded with four different groups. They are named as R and S configuration. For naming the enantiomer as R or S-enantiomer, the Cahn-Ingold-Prelog rule is followed.
(e)
Interpretation:
Whether the following pair of structures is identical molecules, enantiomers or isomers of some other sort:
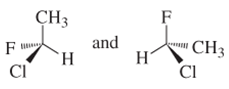
Concept introduction:
Structural or constitutional isomers can be defined as the molecules with same molecular formula and different structural formula. They are molecules in which the bonded atoms have different bonding with each other although the number of molecules is same. Identical molecules are molecules which have same structural formula and same molecular formula.
Enantiomers are optical isomers which can rotate the plane polarized light either clockwise or anticlockwise. These molecules must have at least one chiral C atom which is bonded with four different groups. They are named as R and S configuration. For naming the enantiomer as R or S-enantiomer, the Cahn-Ingold-Prelog rule is followed.
(f)
Interpretation:
Whether the following pair of structures is identical molecules, enantiomers or isomers of some other sort:
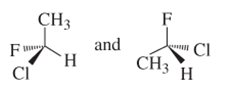
Concept introduction:
Structural or constitutional isomers can be defined as the molecules with same molecular formula and different structural formula. They are molecules in which the bonded atoms have different bonding with each other although the number of molecules is same. Identical molecules are molecules which have same structural formula and same molecular formula.
Enantiomers are optical isomers which can rotate the plane polarized light either clockwise or anticlockwise. These molecules must have at least one chiral C atom which is bonded with four different groups. They are named as R and S configuration. For naming the enantiomer as R or S-enantiomer, the Cahn-Ingold-Prelog rule is followed.
Trending nowThis is a popular solution!

Chapter 26 Solutions
General Chemistry: Principles and Modern Applications - With Solutions Manual and Modified MasteringChemistry Code
- Which of the following molecules can have enantiomers? Identify any chiral carbon atoms. a. b. c.arrow_forwardWhich of the following is the correct relationship between the molecules shown below? (Diastereoisomers, Enantiomers, Identical, Constitutional Isomer, Positional Isomers, Skeletal isomers, Functional Group isomers etc...)arrow_forwardSketch this molecule and then draw a circle around all the chiral carbons that you can find. 1. Classify the type of isomerism (if any) shown by the compounds below.arrow_forward
- A tetrahedral carbon atom will be chiral if it has four ______ groups attached to it Fill in the blankarrow_forwardDetermine if the following compound is chiral or not.arrow_forwardWhat is isomer? Name by typing the isomers of the compound whose closed formula is C4H9Cl. Which of these isomers can have chiral structure. Examine it from a stereochemical point of view.arrow_forward
 Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning
Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning Living By Chemistry: First Edition TextbookChemistryISBN:9781559539418Author:Angelica StacyPublisher:MAC HIGHER
Living By Chemistry: First Edition TextbookChemistryISBN:9781559539418Author:Angelica StacyPublisher:MAC HIGHER Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning



