
Concept explainers
Interpretation:
The structure of triphosphate of guanosine has to be drawn.
Concept Introduction:
Composition of
Sugar: In both DNA and RNA, sugar portion is found. In DNA, the sugar is D-ribose, where at 2’hydroxyl group is absent and in RNA, the hydroxyl group is present at 2’.
Nitrogenous bases: Five types of nitrogenous bases (has unique one-letter code A, G, T, U, and C) are derived from two parent compounds called purine and pyrimidine. The purine derivatives are Adenine and Guanine are two fused nitrogen containing rings. The pyrimidine derivatives are Thymine, Cytosine, and Uracil are only one nitrogen containing six-membered ring. Adenine, Guanine, Thymine, and Cytosine are the nitrogenous bases present in DNA. Adenine, Guanine, Cytosine and Uracil are the nitrogenous bases present in RNA.
Nucleotide: (Nucleoside + phosphate)
Nucleotides are the building blocks of nuclei acids; monomers of DNA and RNA
Nucleoside and its naming: The combination of monosaccharide (sugar) and nitrogenous base is known as nucleoside. The nucleoside names are the nitrogenous base name modified with criteria. While naming nucleoside of purine derivatives the suffix ‘-osine’ is included and for pyrimidine derivatives the suffix ‘-idine’ is used. No prefix used for the nucleosides containing ribose and the prefix ‘deoxy-’ is used for deoxyribose.
Naming nucleotide: At the end of the nucleoside, phosphate group is added. For example, 5’-monophosphate means adding one phosphate group at 5’carbon in the sugar ring. Uridine monophosphate can be written as UMP.
Numbering the atoms in sugar and base rings:
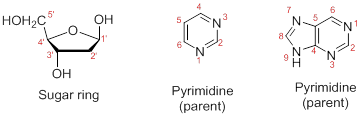
In order to distinguish the atoms in the sugar of a nucleoside and atoms of a base ring, numbers without prime is used for atoms in the base ring and numbers with prime used for the atoms in the sugar ring.
Want to see the full answer?
Check out a sample textbook solution
Chapter 26 Solutions
Fundamentals Of General, Organic And Biological Chemistry In Si Units
- Arrange the following molecules in order of their biological energy content per mole: glucose, caproic acid (C10), sucrose (C12), myristic acid (C14) Write the structural equation for the transamination reaction that involves threonine and pyruvate as pair of reactants [provide the structure of the AA and pyruvate)arrow_forwardList four different products of citric acid cycle.arrow_forward(a) Convert the ball-and-stick model of B, one of the eight synthetic intermediates in the citric acid cycle, to a structural formula, and name the compound. (b) What is the immediate precursor of B in the citric acid cycle; that is, what compound forms B as a reaction product? (c) What compound is formed from B in the citric acid cycle?arrow_forward
- The fact that acid-anhydride bonds are less stable than ester bonds helps explain the fact that the bond between the: Beta- and gamma-phosphoryl groups in atp is a high energy bond, but not the bond between the alpha-phosphorly groups and the hydroxyl of ribose. Phosphorly groups and the hydroxyl at the C-3 position in 3-phosphoglycerate is a high-energy bond. Beta- gamma- phosphoryl groups in ATP is a high-energy bond, but not bond between the alpha and beta-phosphoryl groups. Alpha-phosphorly group (i.e, the one nearest the ribose) and the hydroxyl at the 5’ position in ATP is a high-energy bond.arrow_forwardThere are eight chemical reactions that occur in the citric acid cycle process. The reactions of the citric acid cycle are shown in the figure below. Several classes of organic reactions like these classes include esterification, decarboxylation, hydration reactions, dehydration of alcohols, oxidation of alcohols, oxidation of aldehydes, reduction of aldehydes, and reduction of ketones. Find at least one of these reaction classes in the citric acid cycle. List the reaction number and the reaction. Write a sentence or two on how you were able to make the classification.arrow_forwardShow the reaction mechanism for the catabolism of proline.arrow_forward
- Part A. If you were to expose cells that are undergoing anaerobic respiration to a radioactive carbon isotope in the form of C6H12O6C6H12O6, which of the following molecules would you not expect to be radiolabeled? a)pyruvate b)succinyl CoA c)fructose-1,6-bisphosphate d)lactate Part b.If you were to expose cells that are undergoing aerobic respiration to a radioactive carbon isotope in the form of acetyl-CoA, which of the following molecules would you not expect to be radiolabeled? a)oxaloacetate b)succinyl CoA c)pyruvate d) CO2arrow_forwardWith alanine AA, and using citric acid cycee and glycolysis, which Carbon atom would be labeled 1st with 14C n succinate? Why?arrow_forwardList the products of the citric acid cycle.arrow_forward
- Draw the complete chemical structure of one ATP molecule at physiological pH. (b) In your drawing, label the α-, β- and γ- phosphate groups. (c) Encircle the phosphoanhydride bond that gets cleaved upon ATP hydrolysis.arrow_forwardDraw a six-carbon saturated fatty acid and show where the double bond is created during the first step of b-oxidation. What is the orientation of this bond?arrow_forwardWhich reaction in the citric acid cycle is most analogous to the oxidative decarboxylation of 6- phosphogluconate to ribulose 5-phosphate? What kind of enzymebound intermediate is formed in both reactions?arrow_forward
 BiochemistryBiochemistryISBN:9781319114671Author:Lubert Stryer, Jeremy M. Berg, John L. Tymoczko, Gregory J. Gatto Jr.Publisher:W. H. Freeman
BiochemistryBiochemistryISBN:9781319114671Author:Lubert Stryer, Jeremy M. Berg, John L. Tymoczko, Gregory J. Gatto Jr.Publisher:W. H. Freeman Lehninger Principles of BiochemistryBiochemistryISBN:9781464126116Author:David L. Nelson, Michael M. CoxPublisher:W. H. Freeman
Lehninger Principles of BiochemistryBiochemistryISBN:9781464126116Author:David L. Nelson, Michael M. CoxPublisher:W. H. Freeman Fundamentals of Biochemistry: Life at the Molecul...BiochemistryISBN:9781118918401Author:Donald Voet, Judith G. Voet, Charlotte W. PrattPublisher:WILEY
Fundamentals of Biochemistry: Life at the Molecul...BiochemistryISBN:9781118918401Author:Donald Voet, Judith G. Voet, Charlotte W. PrattPublisher:WILEY BiochemistryBiochemistryISBN:9781305961135Author:Mary K. Campbell, Shawn O. Farrell, Owen M. McDougalPublisher:Cengage Learning
BiochemistryBiochemistryISBN:9781305961135Author:Mary K. Campbell, Shawn O. Farrell, Owen M. McDougalPublisher:Cengage Learning BiochemistryBiochemistryISBN:9781305577206Author:Reginald H. Garrett, Charles M. GrishamPublisher:Cengage Learning
BiochemistryBiochemistryISBN:9781305577206Author:Reginald H. Garrett, Charles M. GrishamPublisher:Cengage Learning Fundamentals of General, Organic, and Biological ...BiochemistryISBN:9780134015187Author:John E. McMurry, David S. Ballantine, Carl A. Hoeger, Virginia E. PetersonPublisher:PEARSON
Fundamentals of General, Organic, and Biological ...BiochemistryISBN:9780134015187Author:John E. McMurry, David S. Ballantine, Carl A. Hoeger, Virginia E. PetersonPublisher:PEARSON





