
Concept explainers
(a)
Interpretation:
To identify the reactant which reacts with dilute hydrochloric acid
Concept introduction:
Nucleophiles are those species which are electron rich in nature. They are negatively charged. Carboxylate ion is a very good nucleophile. So, it can reacts with dilute
(b)
Interpretation:
To identify the reactant that undergoes hydrolysis.
Concept introduction:
The structure of ester is,
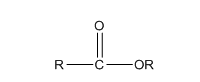
The ester undergoes hydrolysis reaction to form carboxylic acid and alcohol. The hydrolysis reaction of ester can be performed in both acidic and basic medium.
(c)
Interpretation:
To identify the reactant that neutralizses dilute NaOH.
Concept introduction:
The reaction of an acid and base is called neutralization reaction. The products of neutralization reaction are salt and water. So, the reactant that can neutralize
Want to see the full answer?
Check out a sample textbook solution
Chapter 27 Solutions
Generl Chem Looself&mod Mst/et&stdy Crd Pkg, 11/e
- Lipoic acid is required by many microorganisms for proper growth. As a disulfide, it functions in the living system by catalyzing certain oxidation reactions and is reduced in the process. Write the structure of the reduction product.arrow_forwardList the requested biological classification or organic functional group(s) for the reactant, predict the reaction type, and predict the product(s) classification or functional group(s). Use the information to draw the major product(s)arrow_forwardGive the major organic products?arrow_forward
- why is CH3CHCH3 and electrophile and have a + sign and what is its structure and name?arrow_forwardWhich of the following statements about oxidation and reduction is correct? A. Nucleophiles are compounds with high electron donation tendency, they can oxidize easily.B. As a result of the introduction of hydrogen atom into a molecule, an oxidation reaction takes place.A. Oxidizers are compounds with high electron affinity.D. Separation of a hydride ion by catalytic dehydrogenation from a molecule is a reduction reactionE. Removing the oxygen ion from a molecule transforms that molecule into another reduced molecule.arrow_forwardgive the nomenclature or the structure of the following molecules of item #21 to #25.arrow_forward
 Chemistry & Chemical ReactivityChemistryISBN:9781337399074Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning
Chemistry & Chemical ReactivityChemistryISBN:9781337399074Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning Introductory Chemistry: A FoundationChemistryISBN:9781337399425Author:Steven S. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
Introductory Chemistry: A FoundationChemistryISBN:9781337399425Author:Steven S. Zumdahl, Donald J. DeCostePublisher:Cengage Learning Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning
Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning Macroscale and Microscale Organic ExperimentsChemistryISBN:9781305577190Author:Kenneth L. Williamson, Katherine M. MastersPublisher:Brooks Cole
Macroscale and Microscale Organic ExperimentsChemistryISBN:9781305577190Author:Kenneth L. Williamson, Katherine M. MastersPublisher:Brooks Cole



