
ORG CHEM CONNECT CARD
6th Edition
ISBN: 9781264860746
Author: SMITH
Publisher: MCG
expand_more
expand_more
format_list_bulleted
Concept explainers
Textbook Question
Chapter 27, Problem 70P
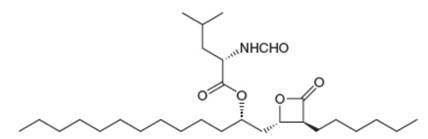
The anti-obesity drug orlistat works by irreversibly inhibiting pancreatic lipase, an enzyme responsible for the hydrolysis of triacylglycerols in the intestines, so they are excreted without
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
Esterase is an enzyme that catalyzes the hydrolysis of esters. It hydrolyzes esters of L-amino acids more rapidly than esters of d-amino acids. How can this enzyme be used to separate a racemic mixture of amino acids?
An enzyme catalyzes the hydrolysis of an ester with a
certain activity, but this activity is lost in a 3 M urea
solution. What is the most likely explanation for the loss
of activity?
(A) Urea binds to the active site of the enzyme
competitively with the substrate.
(B) Urea causes the cleavage of the peptide bonds in the
enzyme.
(C) Urea causes the enzyme to denature and lose its
specific three-dimensional shape.
(D) Urea reacts with disulfide bonds in the enzyme.
Neostigmine is an inhibitor of acetylcholinesterase. The enzyme attempts to catalyse the same reaction on neostigmine as it does with acetylcholine. However, a stable intermediate is formed which prevents completion of the process and which results in a molecule being covalently linked to the active site. Identify (draw) the stable intermediate and explain why it is stable.
Chapter 27 Solutions
ORG CHEM CONNECT CARD
Ch. 27.1 - Prob. 1PCh. 27.1 - Problem 29.2
What form exists at the isoelectric...Ch. 27.1 - Problem 29.3
Explain why the of the group of an...Ch. 27.1 - Prob. 4PCh. 27.2 - Problem 29.5
What -halo carbonyl compound is...Ch. 27.2 - Problem 29.6
The enolate derived from diethyl...Ch. 27.2 - Problem 29.7
What amino acid is formed when is...Ch. 27.2 - Problem 29.8
What aldehyde is needed to synthesize...Ch. 27.2 - Problem 29.9
Draw the products of each...Ch. 27.3 - Prob. 10P
Ch. 27.3 - Prob. 11PCh. 27.3 - Prob. 12PCh. 27.4 - Problem 29.13
What alkene is needed to synthesize...Ch. 27.5 - Problem 29.14
Draw the structure of each peptide....Ch. 27.5 - Problem 29.15
Name each peptide using both the...Ch. 27 - Draw the product formed when the following amino...Ch. 27 - With reference to the following peptide: a...Ch. 27 - Prob. 31PCh. 27 - Histidine is classified as a basic amino acid...Ch. 27 - Tryptophan is not classified as a basic amino acid...Ch. 27 - What is the structure of each amino acid at its...Ch. 27 - What is the predominant form of each of the...Ch. 27 - 29.37 What is the predominant form of each of the...Ch. 27 - a. Draw the structure of the tripeptide A–A–A, and...Ch. 27 - 29.39 Draw the organic products formed in each...Ch. 27 - 29.40 What alkyl halide is needed to synthesize...Ch. 27 - Prob. 50PCh. 27 - Draw the structure for each peptide: (a) Phe–Ala;...Ch. 27 - 29.52 For the tetrapeptide Asp–Arg–Val–Tyr:
a....Ch. 27 - Prob. 53PCh. 27 - Prob. 54PCh. 27 - 29.55 Draw the amino acids and peptide fragments...Ch. 27 - Prob. 56PCh. 27 - Prob. 57PCh. 27 - Prob. 58PCh. 27 - 29.59 An octapeptide contains the following amino...Ch. 27 - 29.60 Draw the organic products formed in each...Ch. 27 - 29.65 Draw the mechanism for the reaction that...Ch. 27 - 29.66 Which of the following amino acids are...Ch. 27 - 29.67 After the peptide chain of collagen has been...Ch. 27 - Prob. 68PCh. 27 - Prob. 69PCh. 27 - 29.70 The anti-obesity drug orlistat works by...Ch. 27 - Prob. 71P
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- The first step in the catabolism of most amino acids is the removal of the nitrogen atom by transfer to an a-keto acid, a reaction catalyzed by an enzyme called a transaminase. The a-keto acid acceptor is often a-ketoglutarate. Modify the structures in the product to show the products of the transamination of cysteine. Be sure to show functional groups with the charge and number of attached hydrogen atoms appropriate for pH 7.4. transaminase + O=C H₂N-CH + CH₂ CH₂ CH₂ SH Incorrect H₂N || CH | CH₂ | CH₂ I || O || n | CH₂ T SHarrow_forwardIt is known that the amino acid at position 523 of the cyclooxygenase enzyme is part of the active site. In the isoenzyme COX-1, this amino acid is isoleucine, whereas in COX-2, it is valine. Suggest how such information could be used in the design of drugs that selectively inhibit COX-2.arrow_forwardTriosephosphate isomerase (TIM) catalyzes the conversion of dihydroxyacetone phosphate to glyceraldehyde-3-phosphate. The enzyme’s catalytic groups are Glu 165 and His 95. In the first step of the reaction, these catalytic groups function as a general-base and a general-acid catalyst, respectively. Propose a mechanism for the reaction.arrow_forward
- Chorismate mutase is an enzyme that promotes a pericyclic reaction by forcing the substrate to assume the conformation needed for the reaction. The product of the pericyclic reaction is prephenate that is subsequently converted into the amino acids phenylalanine and tyrosine. What kind of a pericyclic reaction does chorismate mutase catalyze?arrow_forwardDiazepam is converted in vivo to an active metabolite Oxazepam. By using chemical equations show metabolic transformation of diazepam to oxazepam. Provide name of enzymes involved in these reactions.arrow_forwardChemistry Describe two chemical strategies that have been employed to try to overcome the problem of penicillin-based antibiotics susceptibility to β-Lactamases.arrow_forward
- Acetylcholine esterase is an important enzyme in neural synaptic signal transmission. It breaks down the neurotransmitter acetylcholine after it binds the acetylcholine receptor. Its active site has the same features used by serine proteases. Here is the reaction catalyzed by Acetylcholine esterase: Acetylcholine Choline OH Acetate + H Acetylcholine is an ester, while peptide bonds cleaved by proteases are amides. How should acetylcholine estease's mechanism accommodate this? This cannot be determined from the information presented in class and in this question. For amides, the amide nitrogen is deprotonate to make it more like an ester. The same mechanism cleaves deprotonated amides and esters. No changes are needed. The mechanism same works for esters and amides. Prior to cleavage, a transamination converts the ester to an amide. The esterase will make a covalent intermediate to an amine to create the amide to be cleaved.arrow_forwardlodoacetamide acts as an irreversible inhibitor for a few enzymes by reacting with the amino acid at the active site having the function group -NH_2 -COOH -SH -OHarrow_forwardhi, I'm stuck on these if you can please help Detail the role of each enzyme (five) involved in lipid transport. Enzyme Transporters of Lipids: 1. ABCA1 2. Cholestethin 3. LCAT-phosphotidyl choline 4. ACAT- Transferase 5. Lipoprotein lipase thank youarrow_forward
- 3a. 3b. 3c 3d. 3e. CO₂ clavulanic acid CH₂-OH H Answer the following questions about clavulanic acid. Does clavulanic acid inhibit D-alanyl-D-alanine transpeptidase? Does clavulanic acid contain a ß-lactam? Does clavulanic acid contain a thiazolium ring? What is the result of the treatment of penicillinase with clavulanic acid? Does clavulanic acid form a covalent acyl-enzyme intermediate with penicillinase?arrow_forwardA chemist wanted to test his hypothesis that the disulfide bridges that form in many proteins do so after the minimum energy conformation of the protein has been achieved. He treated a sample of an enzyme that contained four disulfide bridges with 2-mercaptoethanol and then added urea to denature the enzyme. He slowly removed these reagents so that the enzyme could re-fold and re-form the disulfide bridges. The enzyme he recovered had 80% of its original activity. What would be the percent activity in the recovered enzyme if disulfide bridge formation were entirely random rather than determined by the tertiary structure? Does this experiment support his hypothesis?arrow_forwardPlease draw by hand. Triosephosphate isomerase (TIM) catalyzes the conversion of dihydroxyacetone phosphate to glyceraldehyde-3-phosphate. The enzyme's catalytic groups are Glu 165 and His 95. In the first step of the reaction, these catalytic groups function as a base and an acid catalyst, respectively. Propose a mechanism for the reaction. ОН 2-03Р0 ОН dihydroxyacetone phosphate triosephosphate isomerase 2-03РО. H glyceraldehyde-3-phosphate FYI Glu is glutamic acid and his is histadinearrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning
Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning
Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning General, Organic, and Biological ChemistryChemistryISBN:9781285853918Author:H. Stephen StokerPublisher:Cengage Learning
General, Organic, and Biological ChemistryChemistryISBN:9781285853918Author:H. Stephen StokerPublisher:Cengage Learning Organic And Biological ChemistryChemistryISBN:9781305081079Author:STOKER, H. Stephen (howard Stephen)Publisher:Cengage Learning,
Organic And Biological ChemistryChemistryISBN:9781305081079Author:STOKER, H. Stephen (howard Stephen)Publisher:Cengage Learning,

Organic Chemistry
Chemistry
ISBN:9781305580350
Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. Foote
Publisher:Cengage Learning

Introduction to General, Organic and Biochemistry
Chemistry
ISBN:9781285869759
Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar Torres
Publisher:Cengage Learning

Chemistry for Today: General, Organic, and Bioche...
Chemistry
ISBN:9781305960060
Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. Hansen
Publisher:Cengage Learning

General, Organic, and Biological Chemistry
Chemistry
ISBN:9781285853918
Author:H. Stephen Stoker
Publisher:Cengage Learning

Organic And Biological Chemistry
Chemistry
ISBN:9781305081079
Author:STOKER, H. Stephen (howard Stephen)
Publisher:Cengage Learning,

DIGESTER-35 | VITAMINS AND THEIR RELATED COENZYMES| GPAT | NIPER | PHARMACIST| DI; Author: GPAT DISCUSSION CENTER;https://www.youtube.com/watch?v=CGrdNYmho0s;License: Standard YouTube License, CC-BY