
(a)
Interpretation:
It should be identified that will a thermal 1, 3-migrations of carbon occur with whether retention or inversion of configuration.
Concept introduction:
Pericyclic reactions are “ any concerted reaction in which bonds are formed or brocken in a cyclic transition state”. There is a single transition state from start to finish, in contrast to a stepwise reaction.
There are mainly three types of pericyclic reactions,
- 1) Electrocyclic reactions
- 2) Cycloaddition reactions
- 3) Sigmatropic reactions
In a sigmatropic reaction “ one new sigma-bond is formed as another breaks.”
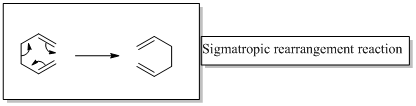
Sigmatropic rearrangement reactions are named with digits. For example a [1, 3] sigmatropic rearrangement describe a reaction in which the residue migrates from position 1 to position 3.
Migration of carbon and hydrogen will occur in a sigmatropic rearrangement reaction.
When hydrogen migrates in a sigmatropic rearrangement, the s orbital of the hydrogen is partially bonded to both the migration origin and the migration terminus in the transition state.
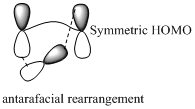
Migration of hydrogen in suprafacial and antarafacial rearrangement can be represented as follows,


Migration of carbon occurs through two ways because it has a two lobed p orbital. Carbon can simultaneously interact with the migration origin and the migration terminus using one lobe of its p orbital.
Migration of carbon in suprafacial and antarafacial rearrangement can be represented as follows,
Carbon migrating with one lobe of its p orbital interacting


Carbon migrating with both lobe of its p orbital interacting

Inversion of configuration is the inversion of a chiral center in a molecule in a
Retention of configuration is the preservation of integrity of the spatial arrangement of bonds to a chiral center during a chemical reaction.
(b)
Interpretation:
It should be identified that will a thermal 1, 5-migrations of carbon occur with whether retention or inversion of configuration.
Concept introduction:
Pericyclic reactions are “ any concerted reaction in which bonds are formed or brocken in a cyclic transition state”. There is a single transition state from start to finish, in contrast to a stepwise reaction.
There are mainly three types of pericyclic reactions,
- 1) Electrocyclic reactions
- 2) Cycloaddition reactions
- 3) Sigmatropic reactions
In a sigmatropic reaction “ one new sigma-bond is formed as another breaks.”

Sigmatropic rearrangement reactions are named with digits. For example a [1, 3] sigmatropic rearrangement describe a reaction in which the residue migrates from position 1 to position 3.
Migration of carbon and hydrogen will occur in a sigmatropic rearrangement reaction.
When hydrogen migrates in a sigmatropic rearrangement, the s orbital of the hydrogen is partially bonded to both the migration origin and the migration terminus in the transition state.
Migration of hydrogen in suprafacial and antarafacial rearrangement can be represented as follows,


Migration of carbon occurs through two ways because it has a two lobed p orbital. Carbon can simultaneously interact with the migration origin and the migration terminus using one lobe of its p orbital.
Migration of carbon in suprafacial and antarafacial rearrangement can be represented as follows,
Carbon migrating with one lobe of its p orbital interacting


Carbon migrating with both lobe of its p orbital interacting


Inversion of configuration is the inversion of a chiral center in a molecule in a chemical reaction.
Retention of configuration is the preservation of integrity of the spatial arrangement of bonds to a chiral center during a chemical reaction.
Want to see the full answer?
Check out a sample textbook solution
Chapter 28 Solutions
Organic Chemistry; Modified MasteringChemistry with Pearson eText -- ValuePack Access Card; Study Guide and Student Solutions Manual for Organic Chemistry, Books a la Carte Edition (7th Edition)
- Which of the following compounds can undergo both T-n* and n-n* excitations? I. 1,3-butadien II. acetone A. I and IV III. Ethanol C. III and IV IV. benzophenone D. Il and IV B. Il and IIIarrow_forwardWhat is the KE of translational androtational motions,in RT units, for(CN)2 and H2O? a.(CN)2 : KE(trans) = 3/2RT & KE(rot) = RT H2O : KE(trans) = 3/2RT & KE(rot) = 3/2RT b.(CN)2 : KE(trans) = 3/2RT & KE(rot) = 3/2RT H2O : KE(trans) = 3/2RT & KE(rot) = 1/2RT c.(CN)2 : KE(trans) = 3/2RT & KE(rot) = 7/2RT H2O : KE(trans) = 3/2RT & KE(rot) = 7/2RT d.(CN)2 : KE(trans) = 3/2RT & KE(rot) = 5/2RT H2O : KE(trans) = 3/2RT & KE(rot) = 3/2RT e.(CN)2 : KE(trans) = 2/2RT & KE(rot) = 5/2RT H2O : KE(trans) = 3/2RT & KE(rot) = 3/2RTarrow_forwardWhat is involved in Phases l and Phases ll of biotransformation?arrow_forward
- True or False: The spin-spin (transverse) relaxation time is the time required for the nuclear magnetic moments to lose phase coherence (the time needed for the magnetic moment to “fan out”)arrow_forward-1 56 The first three Stokes lines in the rotational Raman spectrum of ¹0₂ are separated by 14.4 cm¹, 25.8 cm¹ and 37.4 cm from the exciting radiation. Using the rigid rotor approximation obtain a value for ro.arrow_forward5. Hence show that: OU (hv exp(-ẞhv)) hv exp(-2ẞhv) =-3Nhv + oß (1-exp(-ßhv)) (1-exp(-ẞhv)) Hint 1: you will need to use the quotient rule: yf'(x) xf'(y) f'(x) f'(y) (f'(y))² (f'(y))² Hint 2: the following identity will avoid you having to use the chain rule: d (1 − e√(x)) = − f'(x)e√(x) dxarrow_forward
- Acetaldehyde shows two UV bands, one with a λmax of 289 nm (ε = 12) and one with a λmax of 182 nm (ε = 10,000). Which one is the n -> π* transition and which is the π -> π* transition? Explain your reasoning.arrow_forward1P:18 H.W_cb040d6af52ed4a10c727e8c47881b07.pdf Case 1 A 43-year-old patient suffering from frequent urination and dry tongue was asked by the doctor to analyze RBS, HbA1c and GTT. Results were as follows: RBS = 45 mg\dl 2h GTT = 250 mg\dl HbA1c = 7.5% %3D What interpretation of the result? Case 2 A 35-year-old pregnant woman had a regular check-up and the doctor found the sugar slightly elevated (RBS = 146 mg\dl). What interpretation of the result?arrow_forwardWhich of the following(s) is/ are false ? I- Nonradiative relaxation takes place during collisions between excited molecules and molecules of the solvent. Il- A monochromator enables the spectral isolation of a wavelength from the beam of a light source. III- Vibrational relaxation is the transition between the lower vibrational levels of an excited electronic state and the higher vibrational levels of lower electronic state. Select one: O a. I and ll O b. Il and II O c. only II O d. I and ll O e. l, Il and III O f. only I| O g. only Iarrow_forward
- You add 1 mL of sample and collect 1 mL fractions from a column with a volume of 20 mL where the beads take up 60% of the column and the protein of interest has a partition coefficient of 0.4. What fraction do you want to keep?arrow_forwardBy what factor must the applied magnetic field be increased for the relative population difference (Nɑ - Nβ)/N to be increased by a factor of 5 for (i) 1H nuclei, (ii) 13C nuclei?arrow_forwardChemistry what is the ppm shift predictions for the HNMR of Cr(acac)3.arrow_forward
 Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning EBK A SMALL SCALE APPROACH TO ORGANIC LChemistryISBN:9781305446021Author:LampmanPublisher:CENGAGE LEARNING - CONSIGNMENT
EBK A SMALL SCALE APPROACH TO ORGANIC LChemistryISBN:9781305446021Author:LampmanPublisher:CENGAGE LEARNING - CONSIGNMENT Chemistry for Engineering StudentsChemistryISBN:9781285199023Author:Lawrence S. Brown, Tom HolmePublisher:Cengage Learning
Chemistry for Engineering StudentsChemistryISBN:9781285199023Author:Lawrence S. Brown, Tom HolmePublisher:Cengage Learning


