
Concept explainers
Interpretation:
The differences in the products obtained under photochemical and thermal condition should be accounted for the given reaction.
Concept introduction:
Pericyclic reactions are “ any concerted reaction in which bonds are formed or brocken in a cyclic transition state”. There is a single transition state from start to finish, in contrast to a stepwise reaction.
There are mainly three types of pericyclic reactions,
- 1) Electrocyclic reactions
- 2) Cycloaddition reactions
- 3) Sigmatropic reactions
In a sigmatropic rearrangement reaction “ one new sigma-bond is formed as another breaks.”
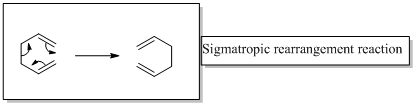
Sigmatropic rearrangement reactions are designated with digits. For example a [1, 3] sigmatropic rearrangement describe a reaction in which the residue migrates from position 1 to position 3.
Woodward – Hoffmann rules for sigmatropic rearrangement reactions are listed below
Want to see the full answer?
Check out a sample textbook solution
Chapter 28 Solutions
Organic Chemistry; Modified MasteringChemistry with Pearson eText -- ValuePack Access Card; Study Guide and Student Solutions Manual for Organic Chemistry, Books a la Carte Edition (7th Edition)
- (b) The rate constant for the solvolysis of X=H, Y=H is 1.14 times greater than that of the dideutero X=D, Y=H but almost identical (0.99) with that of the monodeutero X=H, Y=D. X. Ме,arrow_forwardThe aerobic oxidation of para-xylene to terephthalic acid is an important process in industrial chemistry. Discuss why the oxidation of the second methyl group requires harsher conditions than the oxidation of the first methyl group. You should accurately reference all your bibliographic material.arrow_forwardWhich products are obtained by the following reactions (a) and (b)? Specify the expected main product, if several products can be formed.arrow_forward
- Consider a hypothetical chemical reaction between compound A and compound B, which produces compound C as the final product. The reaction is known to be exothermic and spontaneous. However, when the reaction is carried out under certain conditions, it fails to occur. Explain this observation and propose a potential solution to overcome this hurdle.arrow_forward(b) Explain the possible reagents for the following chemical reactions. I. 2. II. 1. `OH 2.arrow_forwardplease make the explanation clear and thank you for your helparrow_forward
- Suggest a method to prepare ethanol from acetylene ?arrow_forward10. The following reaction has variation of free energy value of AG = -2.1 kJ/mol CH;Br + H2S 2 CH;SH + HBr (a) Calculate Keq at room temperature (25°C) for this reaction as written. (b) Starting with a 1 M solution of CH3B and H2S, calculate the final concentrations of all four species at equilibrium.arrow_forwardWrite the chemical equations involved for the reaction of cyclohexene with cold KMnO4arrow_forward
- A medicinal chemist at Orgalex Labs, a little known division of a big international pharmaceutical firm, has predicted the major product of the following reacti an early step in the synthesis of Compound R40 (rumored to be a cure for rhinitis, spontaneous combustion, and bad luck): or=0x0 HBr OH As Vice-President of Research, it's your job to decide whether the chemist has made a reasonable prediction. • If your answer is yes, these could be the major products of the reaction, draw the complete mechanism for the reaction below. • If your answer is no, check the box under the drawing area. Ö U OH Br H-Br Add/Remove step Click and drag to start drawing a structure.arrow_forward6A Give the major products in the following reactions and explain CN (i) KMnO4 (ii) T. H₂SO4arrow_forwardA proposed mechanism for ozone destruction in the late spring over northern latitudes in the lower stratosphere begins with the photochemical decomposition of CIONO₂ to Cl and NO3, followed by photochemical decomposition of the latter to NO and O₂. Deduce a catalytic ozone destruction cycle, requiring no atomic oxygen.arrow_forward
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY





