
Concept explainers
Interpretation:
It should explained that why was a deuterated compound used in the given reaction.
Concept introduction:
Pericyclic reactions are “ any concerted reaction in which bonds are formed or brocken in a cyclic transition state”. There is a single transition state from start to finish, in contrast to a stepwise reaction.
There are mainly three types of pericyclic reactions,
- 1) Electrocyclic reactions
- 2) Cycloaddition reactions
- 3) Sigmatropic reactions
In a sigmatropic reaction “ one new sigma-bond is formed as another breaks.”
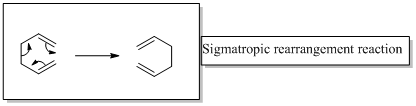
Sigmatropic rearrangement reactions are named with digits. For example a [1, 3] sigmatropic rearrangement describe a reaction in which the residue migrates from position 1 to position 3.
Want to see the full answer?
Check out a sample textbook solution
Chapter 28 Solutions
Organic Chemistry; Modified MasteringChemistry with Pearson eText -- ValuePack Access Card; Study Guide and Student Solutions Manual for Organic Chemistry, Books a la Carte Edition (7th Edition)
- What reagent(s) can be used for that last reaction. Only use ones available in the answer bank please.arrow_forwardWhat is this reaction’s productarrow_forwardCan the alkyl halide in the drawing area be made in one step from an alcohol, without losing starting material to unwanted extra products? If so, add the alcohol starting material to the left-hand side of the reaction, and the necessary reagents above or below the arrow. If the alkyl halide can't be made in one step, or can't be made in good yield because of competing reactions or unwanted byproducts, check the box under the drawing area. Click and drag to start drawing a structure. + X Brarrow_forward
- What's an appropriate reagents/conditions that goes inside the box (step 2)arrow_forwardComplete each reaction by putting the missing reagent or missing product.arrow_forwardDraw both resonance structures of the most stable carbocation intermediate in the reaction shown. + HCI •You do not have to consider stereochemistry. Do not include anionic counter-ions, e.g., I, in your answer. • Draw one structure per sketcher. Add additional sketchers using the drop-down menu in the bottom Separate resonance structures using the → symbol from the drop-down menu. • Draw both resonance structures of the most stable carbocation intermediate in the reaction shown. + HCI • You do not have to consider stereochemistry. • Do not include anionic counter-ions, e.g., I, in your answer. • Draw one structure per sketcher. Add additional sketchers using the drop-down menu in the bottom righ Separate resonance structures using the symbol from the drop-down menu. • - Draw both resonance structures of the most stable carbocation intermediate in the reaction shown. + HBr • You do not have to consider stereochemistry. Do not include anionic counter-ions, e.g., I, in your answer. •…arrow_forward
- reagents for this reactionarrow_forwardShow the steps of the next reaction in detail. And explain why it's hard to go along with the dotted line.arrow_forward6. Circle the structure with the most nucleophilic double bond. Put a box around the structure with the least nucleophilic double bond. OHarrow_forward
- What would be the major product if they are reacted with the reagents in the second column?arrow_forward1. Attack the alkyl chloride with the stronger nucleophile. 2. Deprotonated to form the productarrow_forwardDraw the structure of an alkene with one double bond that would give the following compound as the only product after ozonolysis followed by H,0,. HO HO. Draw the alkene starting material. Erase Select Draw Rings Morearrow_forward
 Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
