
(a)
Interpretation: The mode of ring closure for each of the given electro cyclic reactions has to be identified.
Concept introduction:
In an electrocyclic reaction “one new sigma- bond is formed or brocken.”
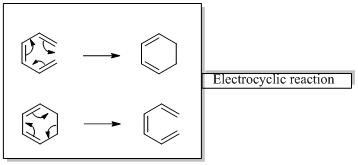
Woodward –Hoffmann rules are the set of rules used to vindicate or predict certain aspects of the stereo chemical outcome and activation energy of pericyclic reactions.
Woodward – Hoffmann rules for Electrocyclic reactions are listed below
Woodward – Hoffmann rules for the configuration of electrocyclic reactions are,
In conrotatory mode both the atomic orbitals of the end groups turn in the same direction.
But in the disrotatory the turning direction is different.
(b)
Interpretation: The configuration of the indicated hydrogens in the reactions has to be predicted.
Want to see the full answer?
Check out a sample textbook solution
Chapter 28 Solutions
Organic Chemistry; Modified MasteringChemistry with Pearson eText -- ValuePack Access Card; Study Guide and Student Solutions Manual for Organic Chemistry, Books a la Carte Edition (7th Edition)
- Which of the following rings is the most strained? cyclopropene; cyclopropane; cyclobutane; cyclopentane; cyclohexane after finding the most strained compound explain why and compare it to the other answer choices, explaining why they do not work as an answer.arrow_forwardName the kind of sigmatropic rearrangement that occurs in each of the following reactions. a. Using arrows, show the electron rearrangement that takes place in each reaction.arrow_forward2) a. 2,3-Di-tert-butyl-1,3-butadiene does not under go Diels-Alder reactions. Explain. b. How can you account for the fact that (Z)-1,3-pentadiene is much less reactive than (E)-1,3- pentadiene in Diels-Alder reaction?arrow_forward
- Classify each of the following cycloadditions and explain why orbital symmetry conservation rules allow or forbid each of the following reactions to occur as a concerted process.arrow_forward6. Answer ALL parts of this question: a) Draw all the molecular orbitals of ethene and buta-1,3-diene, indicating where applicable which are the HOMOS and which are the LUMOS in both the ground state and first excited state. b) Use relevant orbitals from those drawn above to explain why buta-1,3- diene reacts with ethene in a [4+2] cycloaddition reaction under thermal conditions, but if two molecules of ethene are to dimerise in a [2+2] cycloaddition reaction, then the reaction must be irradiated with ultraviolet light. c) Identify compound X in the following reaction (Scheme 4) and draw curly arrow mechanisms that lead to it and then lead from it to the final products. CO, Me .CO, Me heat heat CO,Me СО Ме Scheme 4 d) The reaction shown below (Scheme 5) is another example of a cycloaddition reaction. Identify the number of electrons involved in it, draw a curly arrow mechanism leading to the product and suggest whether this reaction would take place under thermal or photochemical…arrow_forward6. Answer ALL parts of this question: a) Draw all the molecular orbitals of ethene and buta-1,3-diene, indicating where applicable which are the HOMOS and which are the LUMOS in both the ground state and first excited state. b) Use relevant orbitals from those drawn above to explain why buta-1,3- diene reacts with ethene in a [4+2] cycloaddition reaction under thermal conditions, but if two molecules of ethene are to dimerise in a [2+2] cycloaddition reaction, then the reaction must be irradiated with ultraviolet light. c) Identify compound X in the following reaction (Scheme 4) and draw curly arrow mechanisms that lead to it and then lead from it to the final products. CO Me CO₂Me heat heat X CO₂Me CO Me Scheme 4 d) The reaction shown below (Scheme 5) is another example of a cycloaddition reaction. Identify the number of electrons involved in it, draw a curly arrow mechanism leading to the product and suggest whether this reaction would take place under thermal or photochemical…arrow_forward
- 6) The Diels-Alder reaction of anthracene (which is achiral) and maleic anhydride (also achiral) results in the formation of an achiral product. This is not always the case (where two achiral reactants form an achiral product). a) Propose a Diels-Alder reaction where two achiral starting materials result in the formation of a chiral product. b) Would this chiral product be a single enantiomer or a racemic mixture?arrow_forward5. The molecule below is the product of a hetero-Diels-Alder reaction, i.e. one in which one or more of the atoms of the reactants that form the six-membered ring of the product is different than carbon. Circle the hetero-Diels-Alder retron in the product and draw the reactants. cat., -20 °C CC14 HN-arrow_forwardA conjugated diene with an even number of double bonds undergoes conrotatory ring closure underthermal conditions.arrow_forward
- Which of the structures below is the major product obtained from the following Diels-Alder reaction. Note that you will have to re-draw the diene in the s-cis conformation to correctly predict the stereochemistry of the product. ? A B Carrow_forwardFor the Diels-Alder reaction, the product's skeletal structure and configuration at the bridgehead carbons are given (and the given wedge bond and dash bond should not be changed). Complete the structure of the major product. Indicate the stereochemistry, by adding two wedge and two dash bonds, at only the fused-ring stereocenters. A Complete the product and clearly show stereochemistry. H Harrow_forwardD) 22. Which of the following WILL NOT act as a diene reactant in a Diels-Alder reaction? B) OCH3arrow_forward
 Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning

