
Concept explainers
Several resonance structures are possible for each of the following ions. For each, drawthese resonance structures, assign formal charges, and select the resonance structure likely toprovide the best description for the ion.
a. Selenocyanate ion,
b. Thioformate ion,
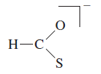
c. Dithiocarbonate,
Learn your wayIncludes step-by-step video

Chapter 3 Solutions
Inorganic Chemistry
Additional Science Textbook Solutions
Organic Chemistry (8th Edition)
Chemistry: Structure and Properties (2nd Edition)
Chemistry: Matter and Change
Chemistry: The Central Science (13th Edition)
Chemistry & Chemical Reactivity
Chemistry: The Central Science (14th Edition)
- Several Lewis structures can be written for perbromate ion, , the central Br with all single Br—O bonds, or with one, two, or three Br=O double bonds. Draw the Lewis structures of these possible resonance structures, and use formal charges to predict which makes the greatest contribution to the resonance hybrid.arrow_forwardWhich of the species with octets in Question 32 are dipoles?arrow_forwardIt is possible to write a simple Lewis structure for the SO42- ion, involving only single bonds, which follows the octet rule. However, Linus Pauling and others have suggested an alternative structure, involving double bonds, in which the sulfur atom is surrounded by six electron pairs. (a) Draw the two Lewis structures. (b) What geometries are predicted for the two structures? (c) What is the hybridization of sulfur in each case? (d) What are the formal charges of the atoms in the two structures?arrow_forward
- The formate ion is HCOO-. Carbon is the central atom. The other atoms are bonded only to the carbon atom. - Draw the best Lewis structure for the formate ion. Show all important resonance structures. Indicate all non-zero formal charges. - In the formate ion, are both carbon-oxygen bonds the same length? What is the oxidation state of carbon in the formate ion? -Which of the following molecules has the shortest carbon-oxygen bond length? HCOO- CO CO2arrow_forwardDraw the Lewis structure of dinitrogen monoxide, N2O, that minimizes formal charges Calculate the electrons required (ER), valence electrons (VE), and shared pairs (SP)arrow_forwardFor the anion IF4- How many total valence electrons should the Lewis structure show? What is the formal charge on I? What is the molecular geometry of this ion? Is the molecule polar?arrow_forward
- The action of dinitrogen on calcium carbide at 1100°C leads to the formation of calcium cyanamideCaNCN following the equation: CaC2 + N2 → CaNCN + C a. What is the charge of the cyanamide ion? b. Write the Lewis structure for the cyanamide ion. All atoms should satisfy the octet rule.Include formal charges if any are present c. By hydrolysis, calcium cyanamide can be transformed into cyanamide with the formulaH2NCN. Write a Lewis structure for cyanamide, knowing that the hydrogen atoms arelocated on the peripheral atoms.arrow_forwardAssign formal charges to each atom in the interhalogen ion ICl4−.arrow_forwardDraw the Lewis structure with lowest formal charges, and determine the chargeof each atom in (i) CN-, (ii) ClO- , (iii) ClNOarrow_forward
- Which of the following would exhibit the greatest amount of covalent bonding (have the lowest differences in electronegativities)? a. FeS b. NO c. SeF2 d. AlNarrow_forwardUse the molar bond enthalpy data in the table to estimate the value of Δ?∘rxnΔHrxn° for the equation NH3(g)+2O2(g)⟶HNO3(g)+H2O(g)NH3(g)+2O2(g)⟶HNO3(g)+H2O(g) The bonding in the molecules is shown.arrow_forwardBased on formal charge considerations, which of the following would likely be the correct arrangement of atoms in hypochlorous acid: HOCl or OClH?arrow_forward
 Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning
Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning
Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning General Chemistry - Standalone book (MindTap Cour...ChemistryISBN:9781305580343Author:Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; DarrellPublisher:Cengage Learning
General Chemistry - Standalone book (MindTap Cour...ChemistryISBN:9781305580343Author:Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; DarrellPublisher:Cengage Learning
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning





