
Inorganic Chemistry
5th Edition
ISBN: 9780321811059
Author: Gary L. Miessler, Paul J. Fischer, Donald A. Tarr
Publisher: Prentice Hall
expand_more
expand_more
format_list_bulleted
Concept explainers
Textbook Question
Chapter 3, Problem 3.39P
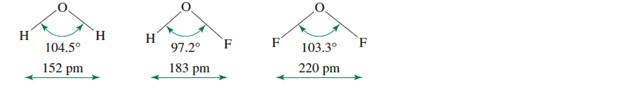
Compounds in which hydrogen is the outer atom can provide challenges to theories ofchemical bonding. Consider the following molecules. Using one or more of the approachesdescribed in this chapter, provide a rationale for HOF having the smallest bond angle in this set.
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
Compounds in which hydrogen is the outer atom can provide challenges to theories of chemical bonding. Consider the following molecules. Using one or more of the approaches described in this chapter, provide a rationale for HOF having the smallest bond angle in this set.
Elaborate on the significance of orbital hybridization and differentiate the different types of orbital hybridization. Also, explain the difference between a sigma bond and pi bond.
( please provide detail )
How will the various d and p orbitals bond to give bonding and anti bonding molecular orbitals. Can you provide diagrams please
Chapter 3 Solutions
Inorganic Chemistry
Ch. 3.1 - Use electron-dot diagrams and formal charges to...Ch. 3.2 - Predict the structures of the following ions....Ch. 3.2 - Prob. 3.3ECh. 3.2 - Which molecule has the smallest bond angle in each...Ch. 3.2 - Briefly account for the following observations: a....Ch. 3.2 - Does this approach work for different steric...Ch. 3 - The dimethyldithiocarbamate ion, [S2CN( CH 3 )2]-...Ch. 3 - Several resonance structures are possible for each...Ch. 3 - Draw the resonance structures for the...Ch. 3 - Three isomers having the formula N2CO are known:...
Ch. 3 - Show the possible resonance structures for nitrous...Ch. 3 - Nitric acid, which exists as HNO3 molecules in the...Ch. 3 - L. C. Allen has suggested that a more meaningful...Ch. 3 - Give Lewis dot structures and sketch the shapes of...Ch. 3 - Give Lewis dot structures and sketch the shapes of...Ch. 3 - Give Lewis dot structures and sketch the shapes of...Ch. 3 - Explain the trends in bond angles and bond lengths...Ch. 3 - Select from each set the molecule or ¡on having...Ch. 3 - a. Compare the structures of the azide ion, N3 ,...Ch. 3 - Consider the series OCl2,O( CH3)2 , and O( SiH3)2...Ch. 3 - Two ions isoelectronic with carbon suboxide, C3O2...Ch. 3 - Explain the following: a. Ethylene, C2H4 , is a...Ch. 3 - Explain the following: a. PCI5 is a stable...Ch. 3 - X-ray crystal structures of ClOF3 and BrOF3 have...Ch. 3 - Make the following comparisons about the molecules...Ch. 3 - Prob. 3.20PCh. 3 - A solution containing the lO2F2 ion reacts slowly...Ch. 3 - The XeOF3 anion has been reported recently (D. S....Ch. 3 - Predict the structure of l(CF3)Cl2 . Do you expect...Ch. 3 - a. Which has the longer axial PF distance, PF2(...Ch. 3 - Prob. 3.25PCh. 3 - SeCl62,TeCl62 , and CIF6 are all octahedral, but...Ch. 3 - Prob. 3.27PCh. 3 - The thiazyldichloride ion, NSCl2- , is...Ch. 3 - Sketch the most likely structure of PCl3Br2 and...Ch. 3 - a. Are the CF3 groups in PCl3( CF3)2 more likely...Ch. 3 - Of the molecules C1SO2CH3,C1SO2CF3 , and ClSO2CCl3...Ch. 3 - Prob. 3.32PCh. 3 - Prob. 3.33PCh. 3 - Prob. 3.34PCh. 3 - Prob. 3.35PCh. 3 - Although the CF distances and the FCF bond angles...Ch. 3 - The Cl...Cl distance in CCl4 is 289 pm, and the...Ch. 3 - The FCF angle in F2CO , shown here, is 109.5°; the...Ch. 3 - Compounds in which hydrogen is the outer atom can...Ch. 3 - For each of the following bonds, indicate which...Ch. 3 - Give Lewis dot structures and shapes for the...Ch. 3 - Give Lewis dot structures and sketch the shapes...Ch. 3 - Which of the molecules in Problem 3.41 are polar?Ch. 3 - Which of the molecules in Problem 3.42 are polar?Ch. 3 - Prob. 3.45PCh. 3 - Prob. 3.46P
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- Give the Lewis-structure of a molecule containingboth a σp-p and a πp-p overlap of orbitals. Thenindicate the σ- en π-bonds in the molecule.arrow_forwardIn the LCAO method, the energy versus separation for a diatomic molecule revealed that a bonding wave function resulted in a deep minimum in the potential energy, resulting in an equilibrium state. The energy-separation curve has no minimum for the antibonding wave function. Explain qualitatively how the charge distribution (or 1912 distribution, if you like) differs between the bonding and anti-bonding configurations and why the bonding configuration is more stable. That is, where and why is the electron density located?arrow_forwardQuestion:What is the significance of the LUMO (Lowest Unoccupied Molecular Orbital) in organic chemistry, and how does it contribute to chemical reactions?arrow_forward
- I already answered the first part which was satisfying the balances filling in the double and triple bonds as well as the unshared electron pairs, as you can see in the second picture I have written those answers and pin. I’m having trouble with converting the Lewis structure into bondline structures if you could please help me out I would appreciate it.arrow_forwardWhat is the name of the following perovskite; KMnF3 Give some theory behind the formation of this perovskite and its structure.arrow_forwardOn the basis of Molecular Orbital Theory argument, account for the reason why the enthalpy of dissociation of N2 molecules is higher than for B2. (Show all needed detail on both diagrams.arrow_forward
- How much of heat is produced when 125 g of acetylene undergoes combustion? Information given ( std enthapy of combustion of acetylene is 1301 KJ) (Some places I have seen this being solved using bond dissociation enthalpy, however the simple moles method is what I would prefer) Thanksarrow_forwardSketch a MO energy level diagram for cluster bonding in E6 in Oh Point group using these orbitals ( A1g, t2g, t1u) and populate it with electrons to give maximum bonding character.If E = a Group 13 element with all valence electrons available for cluster bonding, what would be the charge on the cluster to maximise the bonding interactions?arrow_forwardGive structure , apply ean with complete work to CpReBr2 (sc2h4s)arrow_forward
- There is persuasive evidence for the brief existence of theunstable molecule OPCl.(a) Draw a Lewis diagram for this molecule in which theoctet rule is satisfied on all atoms and the formalcharges on all atoms are zero .(b) The compound OPCl reacts with oxygen to giveO2PCl. Draw a Lewis diagram of O2PCl for which allformal charges are equal to zero. Draw a Lewis diagram in which the octet rule is satisfied on all atoms.arrow_forwardBoth cis and trans isomers of IO2F4- have been observed. Can IR spectra distinguish between these? Explain, supporting your answer on the basis of group theory.arrow_forwardGive the name and the configuration of the following substance bellowarrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 Physical ChemistryChemistryISBN:9781133958437Author:Ball, David W. (david Warren), BAER, TomasPublisher:Wadsworth Cengage Learning,
Physical ChemistryChemistryISBN:9781133958437Author:Ball, David W. (david Warren), BAER, TomasPublisher:Wadsworth Cengage Learning, Principles of Modern ChemistryChemistryISBN:9781305079113Author:David W. Oxtoby, H. Pat Gillis, Laurie J. ButlerPublisher:Cengage Learning
Principles of Modern ChemistryChemistryISBN:9781305079113Author:David W. Oxtoby, H. Pat Gillis, Laurie J. ButlerPublisher:Cengage Learning

Physical Chemistry
Chemistry
ISBN:9781133958437
Author:Ball, David W. (david Warren), BAER, Tomas
Publisher:Wadsworth Cengage Learning,

Principles of Modern Chemistry
Chemistry
ISBN:9781305079113
Author:David W. Oxtoby, H. Pat Gillis, Laurie J. Butler
Publisher:Cengage Learning
Stoichiometry - Chemistry for Massive Creatures: Crash Course Chemistry #6; Author: Crash Course;https://www.youtube.com/watch?v=UL1jmJaUkaQ;License: Standard YouTube License, CC-BY
Bonding (Ionic, Covalent & Metallic) - GCSE Chemistry; Author: Science Shorts;https://www.youtube.com/watch?v=p9MA6Od-zBA;License: Standard YouTube License, CC-BY
General Chemistry 1A. Lecture 12. Two Theories of Bonding.; Author: UCI Open;https://www.youtube.com/watch?v=dLTlL9Z1bh0;License: CC-BY