
Concept explainers
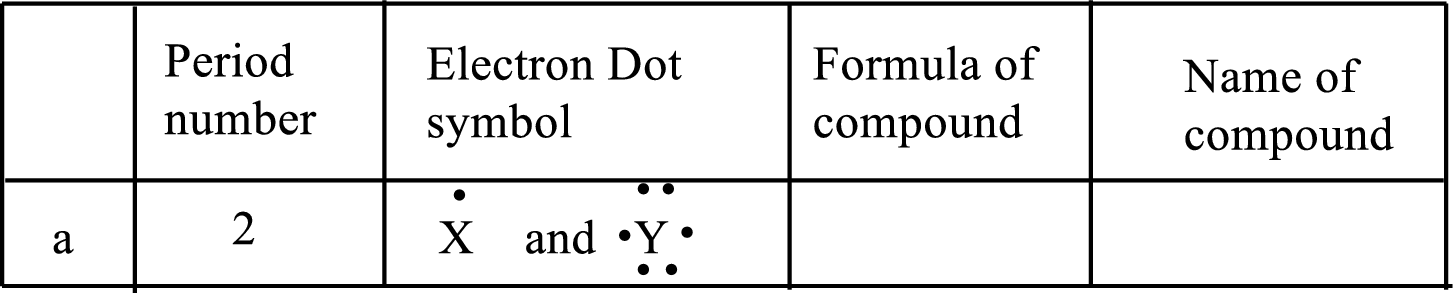
(a)
Interpretation:
Each line of the following table that involve the representative elements
Concept Introduction:
An ionic compound has two elements one is metal and another one is non-metal. The metal ion always has positive charge and the nonmetal ion always has negative ion in binary compounds. Example:
The following rule can be used for the naming of binary ionic compounds.
The full name of the metallic element is given first, followed by a separate word containing the stem of the metallic element name and the suffix –ide.
The positive charge on metal ions from IA, IIA and IIIA is equal to the group number, while the negative charge on non-metal ions from VA, VIA and VIIA is equal to the group number eight.
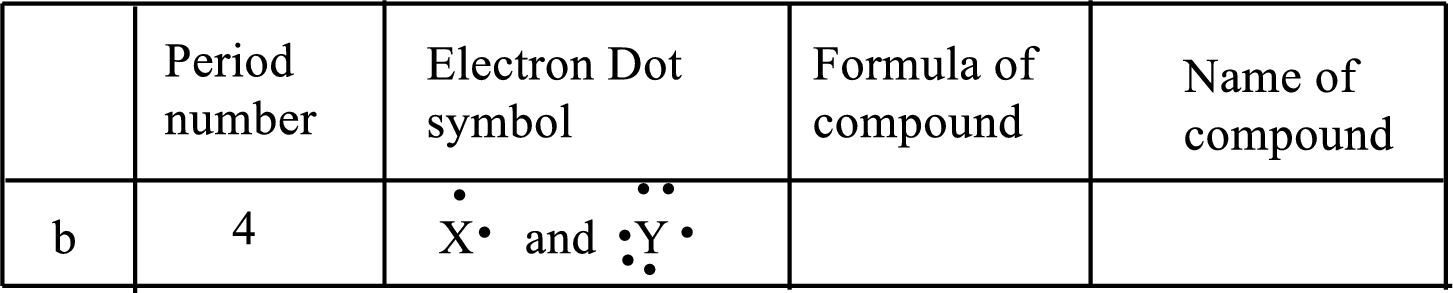
(b)
Interpretation:
Each line of the following table that involve the representative elements
Concept Introduction:
An ionic compound has two elements one is metal and another one is non-metal. The metal ion always has positive charge and the nonmetal ion always has negative ion in binary compounds. Example:
The following rule can be used for the naming of binary ionic compounds.
The full name of the metallic element is given first, followed by a separate word containing the stem of the metallic element name and the suffix –ide.
The positive charge on metal ions from IA, IIA and IIIA is equal to the group number, while the negative charge on non-metal ions from VA, VIA and VIIA is equal to the group number eight.
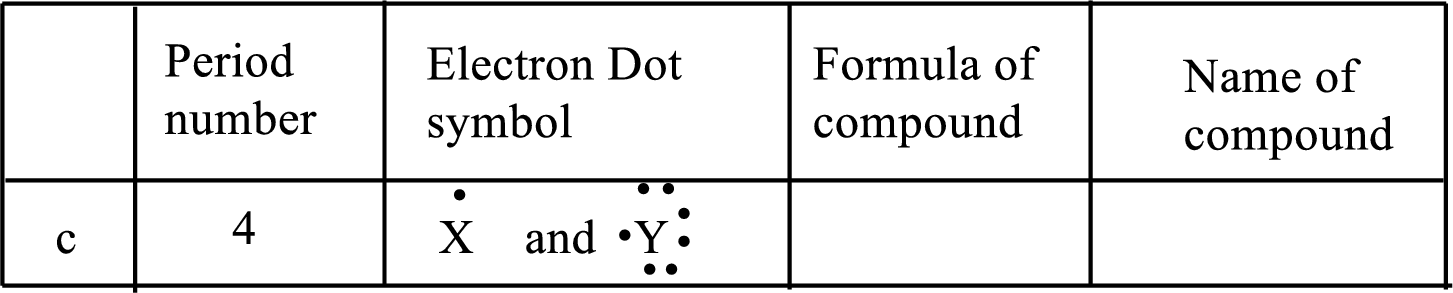
(c)
Interpretation:
Each line of the following table that involve the representative elements
Concept Introduction:
An ionic compound has two elements one is metal and another one is non-metal. The metal ion always has positive charge and the nonmetal ion always has negative ion in binary compounds. Example:
The following rule can be used for the naming of binary ionic compounds.
The full name of the metallic element is given first, followed by a separate word containing the stem of the metallic element name and the suffix –ide.
The positive charge on metal ions from IA, IIA and IIIA is equal to the group number, while the negative charge on non-metal ions from VA, VIA and VIIA is equal to the group number eight.
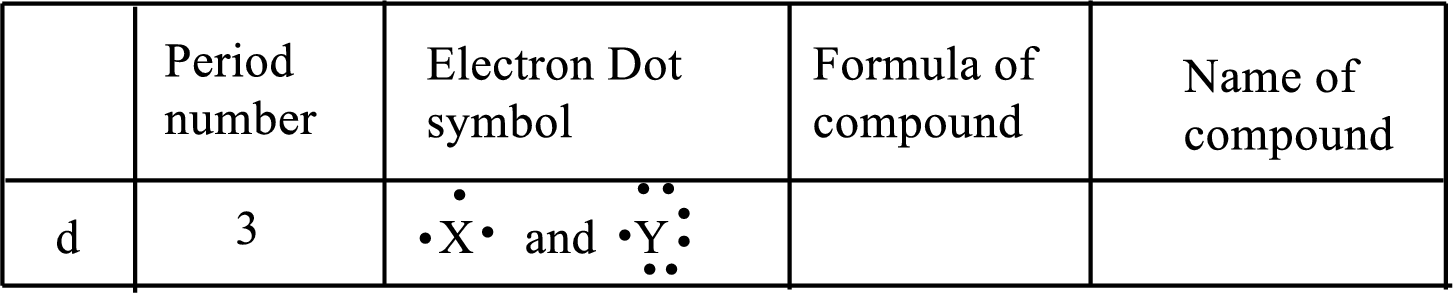
(d)
Interpretation:
Each line of the following table that involve the representative elements
Concept Introduction:
An ionic compound has two elements one is metal and another one is non-metal. The metal ion always has positive charge and the nonmetal ion always has negative ion in binary compounds. Example:
The following rule can be used for the naming of binary ionic compounds.
The full name of the metallic element is given first, followed by a separate word containing the stem of the metallic element name and the suffix –ide.
The positive charge on metal ions from IA, IIA and IIIA is equal to the group number, while the negative charge on non-metal ions from VA, VIA and VIIA is equal to the group number eight.
Want to see the full answer?
Check out a sample textbook solution
Chapter 4 Solutions
General, Organic, and Biological Chemistry
- What are bus? How are ions formed from atoms? Do isolated atoms form ions spontaneously? To what do the termscationandanionrefer? In terms of subatomic particles, how is an ion related to the atom from which it is formed? Does the nucleus of an atom change when the atom is converted into an ion? How can the periodic table be used to predict what ion an element’s atoms will form?arrow_forwardDistinguish between the following terms. a. molecule versus ion b. covalent bonding versus ionic bonding c. molecule versus compound d. anion versus cationarrow_forwardWrite chemical formulas for the following binary ionic compounds. a. Gallium nitride b. Zinc chloride c. Magnesium sulfide d. Aluminum nitridearrow_forward
- Write chemical formulas for the following binary ionic compounds. a. Iron(III) oxide b. Iron(II) oxide c. Nickel(III) sulfide d. Copper(I) bromidearrow_forward2.61 Name the following covalent compounds: (a) N2O5, (b) S2Cl2, (c) NBr3, (d) P4O10arrow_forwardName the following binary ionic compounds, each of which contains a fixed-charge metal. a. CaCl2 b. Ca2C c. Be3N2 d. K2Sarrow_forward
 General, Organic, and Biological ChemistryChemistryISBN:9781285853918Author:H. Stephen StokerPublisher:Cengage Learning
General, Organic, and Biological ChemistryChemistryISBN:9781285853918Author:H. Stephen StokerPublisher:Cengage Learning World of Chemistry, 3rd editionChemistryISBN:9781133109655Author:Steven S. Zumdahl, Susan L. Zumdahl, Donald J. DeCostePublisher:Brooks / Cole / Cengage Learning
World of Chemistry, 3rd editionChemistryISBN:9781133109655Author:Steven S. Zumdahl, Susan L. Zumdahl, Donald J. DeCostePublisher:Brooks / Cole / Cengage Learning Introductory Chemistry: A FoundationChemistryISBN:9781337399425Author:Steven S. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
Introductory Chemistry: A FoundationChemistryISBN:9781337399425Author:Steven S. Zumdahl, Donald J. DeCostePublisher:Cengage Learning Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning
Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning Principles of Modern ChemistryChemistryISBN:9781305079113Author:David W. Oxtoby, H. Pat Gillis, Laurie J. ButlerPublisher:Cengage Learning
Principles of Modern ChemistryChemistryISBN:9781305079113Author:David W. Oxtoby, H. Pat Gillis, Laurie J. ButlerPublisher:Cengage Learning





