
Concept explainers
Artemisinin and mefloquine are widely used antimalarial drugs. A ball-and-stick model of artemisinin appears on the cover of this text.
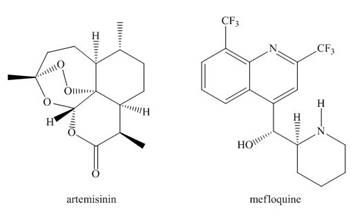
a. Locate the stereogenic centers in both drugs.
b. Label each stereogenic center in mefloquine as
c. What is the maximum number of stereoisomers possible for artemisinin?
d. How are the
e. Can two molecules of artemisinin intermolecularly hydrogen bond to each other?
f. What product is formed when mefloquine is treated with
Trending nowThis is a popular solution!

Chapter 5 Solutions
Organic Chemistry-Package(Custom)
Additional Science Textbook Solutions
Organic Chemistry
Principles of General, Organic, Biological Chemistry
Chemistry: The Central Science (14th Edition)
Chemistry
Chemistry by OpenStax (2015-05-04)
Thermodynamics, Statistical Thermodynamics, & Kinetics
- a. Which of these two can exist as a pair of enantiomers?b. Draw the pair of enantiomers using three-dimensional representation.arrow_forwardConsider the ball-and-stick model of D, and label E and F as either identical to D or an enantiomer of Darrow_forwardLabel the stereogenic center in attached compound as R or S.arrow_forward
- Which compund is optically inactive?arrow_forwardHow is compound A related to compounds B–E? Choose from enantiomers, diastereomers, constitutional isomers, or identical molecules.arrow_forwardTrabectedin, shown in a ball-and-stick model on the cover of this text, isan anticancer drug sold under the trade name Yondelis. Question: a.) Draw the enantiomer. b.) Draw a diastereomer.arrow_forward
- Shown below is Streptomycin, and Neomycin B. Circle and label as many functional groups in these molecules as you can. a. Label each chiral carbon in Streptomycin. How many total stereoisomers exist for Streptomycin? b. Label each chiral carbon in Neomycin B. How many total stereoisomers exist for Neomycin B?arrow_forwardLabel each stereogenic center in the attached compounds as R or S.arrow_forwardCaptopril is a drug used to treat high blood pressure and congestiveheart failure.Draw the enantiomer of captoprilarrow_forward
- How is each compound (B–D) related to A? Choose from enantiomers, diastereomers, identical molecules, constitutional isomers, or not isomers of each other.arrow_forwardStereoisomers share the same connectivity and differ only in the way their atoms are arranged in space. Draw the structure of a compound that is a stereoisomer ofarrow_forwardThe purine heterocycle occurs commonly in the structure of DNA.a.How is each N atom hybridized? b.In what type of orbital does each lone pair on a N atom reside? c.How many π electrons does purine contain? d.Why is purine aromatic?arrow_forward
 Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning
Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning


