
Concept explainers
Under physiological conditions, the protein hemerythrin exists as an octamer of eight chains of the kind shown in Problem 3.
a. Name two symmetries possible for this molecule
b. Which do you think is more likely? Explain.
c. For the more likely symmetry, what kinds of interactions (isologous, heterologous, or both) would you expect? Why?
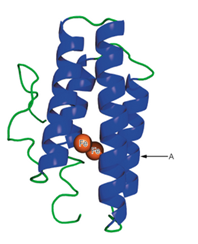
3. A schematic structure of the subunit of hemerythrin (an oxygen-binding protein from invertebrate animals) is shown to the right.
a. It has been found that in some of the a-helical regions of hemerythrin, about every third or fourth amino acid residue is a hydrophobic one. Suggest a structural reason for this finding.
b. What would be the effect of a mutation that placed a proline residue at point A in the structure?
Want to see the full answer?
Check out a sample textbook solution
Chapter 6 Solutions
Biochemistry: Concepts and Connections (2nd Edition)
- In the Watson-Crick DNA base pairing model, Adenine (A) binds to thymine (T), guanine (G) binds to cytosine (C).Draw the structures of thymine and adenine stabilized by Watson-Crick base pair interaction. Also draw the structure of the amide group of glutamine in an interaction of this T-A pair in a way that maximally satisfies the hydrogen bonding capacity of amide.arrow_forwardAs an example, consider albumin, a protein made of a single polypeptide weighing 66,000 daltons (66 kDa). On the other hand, gamma globulin has quaternary structure: is made of four polypeptides, two of which weigh 23,000 daltons (23 kDa) each, and two of which weigh 53,000 daltons (53 kDa) each. When treated with reducing agent and SDS, the subunits separate and they all linearize. If albumin and gamma globulin were run through gel electrophoresis, which polypeptides would move the fastest? Which would move the slowest?arrow_forwardYou are studying the toxic protein called ectatomin, a major component of ant venom (specifically from the species Ectatomma tuberculatum), that embeds into cell membranes and creates pores that cause cells to lyse. Ectatomin is a small dimer, meaning the protein is comprised of two polypeptides; the sequences of the two polypeptides are given in the table with single-letter abbreviations. polypeptide sequences of ectatomin monomers polypeptide protein sequence polypeptide 1 GVIPKKIWETVCPTVEPWAKKCSGDIATYIKRECGKL polypeptide 2 WSTIVKLTICPTLKSMAKKCEGSIATMIKKKCDK You could use absorbance at 280?? or colorimetric assays like the Bradford assay or bicinchoninic acid assay (BCA) assay to quantify the amount of ectatomin you have for your experiment. Why might you choose to use a colorimetric method instead of absorbance at 280??? Select the best answer. Measuring absorbance at higher wavelengths, like 595?? or 562??, is more accurate than measuring absorbance at…arrow_forward
- Avidin exists as a protein complex of around 68 kDa. Research to determine the types of interactions that hold the avidin complex together. Based on your research, which molecular weight would you expect for avidin when treated with beta-mercaptoethanol and run through a native gel? 35 kDa 68 kDa 10 kDa 19 kDaarrow_forwardConsider an a-helix secondary structure.a. Propose a 12-residue amino acid sequence that(assuming it folded intoan a-helix) would yield a structure in which six of the residues, each with an ionizable side-chain were within a 135° region on one face of the helix, and use an appropriate diagram to support your choice of sequence.b. Would it be possible to generate an especially stable helix by correctly choosing the charges on the side-chains? Please justify your answer.arrow_forwardDraw Ramachandran plot for: a) regular secondary structure with Φ = 60-65 and Ψ = 60-80 b) regular secondary structure with Φ = -170 and Ψ = 170 c) intrinsically disordered proteins d) explain why these Ramachsndran plots will be different, and what secondary structures are described in A and in Barrow_forward
- . Give two reasons to explain why a proline residue in the middle of an ahelix is predicted to be destabilizing to the helical structurearrow_forwardA novel sugar "D-Lasallose' was isolated from a native edible mountain mushroom by a group of DLSU-D Biochemistry researchers under Sir Gids. Given the Fischer projection structure of D-Lasallose below,show the systematic conversion into the equivalent Haworth structures and give the systematic names of both anomers.arrow_forwardPectin, which occurs in plant cell walls, exist in nature as a polymer of D-galacturonic acid methylated at carbon 6 of the monomer. Draw a haworth projection for a repeating disaccharide unit of pectin with one methylated and one unmethylated monomer unit in a (1-->4) linkagearrow_forward
- Give two reasons to explain why a proline residue in the middle of an αhelix is predicted to be destabilizing to the helical structure.arrow_forwardLet’s consider histidine as a free amino acid in aqueous solution. a) Draw the most likely structure of histidine under biochemical standard state conditions. b) Given that free histidine has the following three pKa values, assign each to its corresponding acidic hydrogen or conjugate base in your structure from part a). pKa1 = 1.7; pKa2 = 6.2; and pKa3 = 9.1 c) For each pKa, give the corresponding expression for the equilibrium constant. It helps to write out the chemical equation for each. d) Create a speciation diagram for histidine by plotting Xi vs pH from pH = 4 to pH = 8 where Xi is the mole fraction of the two histidine species involved in the K2 equilibrium in part c).arrow_forwardThe ends of eukaryotic chromosomes terminate in a G-rich singlestrandedoverhang that can fold up on itself to form a four-stranded structure. In this structure, four guanine residues assume a hydrogen-bonded planar arrangement with an overall geometry that can be represented as shown below. Draw the complete structure of this “G quartet,” including the hydrogen bonds between the purine bases.arrow_forward
 BiochemistryBiochemistryISBN:9781319114671Author:Lubert Stryer, Jeremy M. Berg, John L. Tymoczko, Gregory J. Gatto Jr.Publisher:W. H. Freeman
BiochemistryBiochemistryISBN:9781319114671Author:Lubert Stryer, Jeremy M. Berg, John L. Tymoczko, Gregory J. Gatto Jr.Publisher:W. H. Freeman Lehninger Principles of BiochemistryBiochemistryISBN:9781464126116Author:David L. Nelson, Michael M. CoxPublisher:W. H. Freeman
Lehninger Principles of BiochemistryBiochemistryISBN:9781464126116Author:David L. Nelson, Michael M. CoxPublisher:W. H. Freeman Fundamentals of Biochemistry: Life at the Molecul...BiochemistryISBN:9781118918401Author:Donald Voet, Judith G. Voet, Charlotte W. PrattPublisher:WILEY
Fundamentals of Biochemistry: Life at the Molecul...BiochemistryISBN:9781118918401Author:Donald Voet, Judith G. Voet, Charlotte W. PrattPublisher:WILEY BiochemistryBiochemistryISBN:9781305961135Author:Mary K. Campbell, Shawn O. Farrell, Owen M. McDougalPublisher:Cengage Learning
BiochemistryBiochemistryISBN:9781305961135Author:Mary K. Campbell, Shawn O. Farrell, Owen M. McDougalPublisher:Cengage Learning BiochemistryBiochemistryISBN:9781305577206Author:Reginald H. Garrett, Charles M. GrishamPublisher:Cengage Learning
BiochemistryBiochemistryISBN:9781305577206Author:Reginald H. Garrett, Charles M. GrishamPublisher:Cengage Learning Fundamentals of General, Organic, and Biological ...BiochemistryISBN:9780134015187Author:John E. McMurry, David S. Ballantine, Carl A. Hoeger, Virginia E. PetersonPublisher:PEARSON
Fundamentals of General, Organic, and Biological ...BiochemistryISBN:9780134015187Author:John E. McMurry, David S. Ballantine, Carl A. Hoeger, Virginia E. PetersonPublisher:PEARSON





