
Biochemistry: Concepts and Connections (2nd Edition)
2nd Edition
ISBN: 9780134641621
Author: Dean R. Appling, Spencer J. Anthony-Cahill, Christopher K. Mathews
Publisher: PEARSON
expand_more
expand_more
format_list_bulleted
Concept explainers
Textbook Question
Chapter 6, Problem 13P
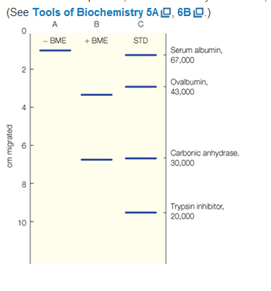
A protein gives under conditions of buffer composition, pH, and temperature that are close to physiological conditions, a molecular weight by size exclusion measurements of 140,000 g/mol. When the same protein studied by SDS gel electrophoresis in the absence or presence of the reducing agent
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
A protein gives, under conditions of buffer composition, pH, and temperaturethat are close to physiological conditions, a molecular weight by size exclusion measurements of 140,000 g/mol. When the same protein is studiedby SDS gel electrophoresis in the absence or presence of the reducing agent β-mercaptoethanol (BME), the patterns seen, respectively, in lanes A and B are observed. Lane C contains standards of molecular weight indicated. From these data, describe the native protein, in terms of the kinds of subunits present, the stoichiometry of subunits, and the kinds of bonding(covalent, noncovalent) existing between subunits.
A protein has a molecular mass of 400 kDa when measured by size-exclusion chromatography. When subjected to gel electrophoresis in the presence of sodium dodecyl sulfate (SDS), the protein gives three bands with molecularmasses of 180, 160, and 60 kDa. When electrophoresis is carried out in the presence of SDS and dithiothreitol, three bands are again formed, this time with molecular masses of 160, 90, and 60 kDa. Determine the subunit composition of the protein.
You are given a pure protein sample to characterize and provided the following information:
Its molar extinction coefficient, ε280, is 0.25 liters micromole-1 cm-1 in both the folded and unfolded form
Its ΔGo for unfolding is 1.5 kcal/mol at 37o (where RT = 0.59 kcal/mole)
A) Using a 0.5 cm pathlength cell, you measure the absorbance at 280 nm of a 20-fold dilution of your pure protein in solution (by this, we mean that 50 ul of the protein sample was diluted to a final volume of 1 ml) and find A280 = 0.40. What is the original concentration of the protein before dilution?
B) What is the concentration of the unfolded form of the protein in your sample?
Chapter 6 Solutions
Biochemistry: Concepts and Connections (2nd Edition)
Ch. 6 - Prob. 1PCh. 6 - Bovine pancreatic trypsin inhibitor (BPTI; Figure...Ch. 6 - A schematic structure of the subunit of...Ch. 6 - In the protein adenylate kinase, the C-terminal...Ch. 6 - Give two reasons to explain why a proline residue...Ch. 6 - Consider a small protein containing 101 amino acid...Ch. 6 - a. Based on a more conservative answer to Problem...Ch. 6 - The following sequence is part of a globular...Ch. 6 - a. A protein is found to be a tetramer of...Ch. 6 - Under physiological conditions, the protein...
Ch. 6 - Theoretical and experimental measurements show...Ch. 6 - The peptide hormone vasopressin is used in the...Ch. 6 - A protein gives under conditions of buffer...Ch. 6 - A protein gives a single band on SDS get...Ch. 6 - It has been postulated that the normal...Ch. 6 - Below are shown two views of the backbone...Ch. 6 - Do you expect a Pro Gly mutation in a...Ch. 6 - Rank the following in terms of predicted rates...Ch. 6 - Shown below are two cartoon views of the small...Ch. 6 - Prob. 20PCh. 6 - In most cases, mutations in the core of protein...Ch. 6 - A Leu Ala mutation at a site buried the core of...Ch. 6 - Disulfide bonds have been shown to stabilize...Ch. 6 - Cartoon renderings of the proteins Top 7 and adaH2...
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, biochemistry and related others by exploring similar questions and additional content below.Similar questions
- Please draw titration curves for peptide HFDTYA and HEKDGQ, and calculate the pl for each. Usethis information to explain why the peptide HFDTYA has low solubility and HEKDGQ has a highersolubility in bulk water at pH = 7.arrow_forwardThis SDS-PAGE loading buffer has SDS (a denaturing detergent), β-mercaptoethanol (a reducing agent), Coomassie Blue (a dye), and glycerol. You will need to dilute your protein samples such that the final concentration of the loading buffer is 1x, meaning that you will add 5 parts of the protein sample for every 1 part of 6x buffer. Calculate how much of the 6x loading buffer you will need to add to each sample. Then, add the appropriate amount of 6x SDS-PAGE loading buffer to the samples. Store the samples in the -20°C freezer. What is the final concentration of glycerol in each of the samples?arrow_forwardA mixture of proteins having molecular weights of 50 to 200 kDa can be fractionated linearly using a GFC column with an exclusion limit of at least _____.a. 25 kDab. 50 kDac. 125 kDad. 200 kDapls explain whyarrow_forward
- The protein concentration of a known standard is 100mg/mL If you prepared a serial dilution, mixing 10μL of protein with 40μL of water what would be concentrations of the first 3 dilutions?arrow_forwardcalculate the volume of stock solutions required to make up the buffer solutions that will be used for protein purification. The solutions you need to prepare for purification are: i. Binding Solution A: make up 50 mL 50 mM HEPES buffer (pH 7.5), 300 mM NaCl, 5mM imidazole, 5% (v/v) glycerol ii. Wash Solution B: make up 50 mL 50 mM HEPES buffer (pH 7.5), 300 mM NaCl, 75mM imidazole, 5% (v/v) glycerol iii. Elution Solution C: make up 10 mL 50 mM HEPES buffer (pH 7.5), 300 mM NaCl, 500 mM imidazole, 5% (v/v) glycerol please show your working . Thnk youarrow_forwardCan you describe the solubility of prolamin, a protein in rice protein bodies, in isopropyl or ethyl alcohol based on its molecular structure?arrow_forward
- A new protein of unknown structure has been purified. Gel filtration chromatography reveals that the native protein has a molecular weight of 300,000. Chromatography of the protein in the presence of 6 M guanidine hydrochloride (a denaturant) yields a single peak corresponding to a molecular weight of 50,000. Chromatography of the protein in the presence of 6 M guanidine hydrochloride and 10 mM beta-mercaptoethanol (a disulfide bond reductant) yields peaks corresponding to molecular weights of 30,000 and 20,000. What does this data tell you about the structure of this protein? (Be thorough in your answer!)arrow_forwardThe vast majority of structures deposited in the Protein Databank (>95%) have been determined using NMR Hard keratins (such as in the skin) are less resistant to stretch because the larger number of disulfide bonds resist deformative forces. Structures produced by X-ray diffraction are as good as the solutions from which they are derived they are false and I know that but explain why they are falsearrow_forwardConsider the following properties of the protein components of a sample mixture as provided in the table below: 1. if the mixture is subjected to gel filtration chromotography which protein component elute first? 2. if the mixture is subjected to isoelectric focusing which protein will stop m oving nearest to the positive electrode? 3. if the mixture is subjected to cation-exchange chromotography using a buffer at ph 7 which protein will bind to the resin? 4.if the mixture is subjected to SDS-PAGE which protein will be at bottomost portion of gel? 5.if the mixture is subjected to hydrophobic interaction chromotography which protein will bind most strongly to the resin?arrow_forward
- A protein has molecular mass of 200 kDa when measured by gel filtration. When subjected to SDS PAGE with and without 2-mercaptoethanol (2-ME) the gel shown below was obtained. What is the likely subunit composition of this protein and why? a. The protein has 4 subunits, with molecular masses 100, 50, 25, and 25 kDa. 25 kDa subunits are linked to each other via noncovalent interactions. b. The protein has 4 subunits, with molecular masses 100, 50, 25, and 25 kDa. 50 kDa subunit is linked to the one 25 kDa subunit via noncovalent interactions. c. The protein has 3 subunits, with molecular masses 100, 75 and 25 kDa that are linked by noncovalent interactions. d. The protein has 4 subunits, with molecular masses 100, 75, 50 and 25 kDa. The subunits are linked by disulfide bonds. e. The protein has 4 subunits, with molecular masses 100, 50, 25, and 25 kDa. 50 kDa subunit is linked to the one 25 kDa subunit by disulfide bonds.arrow_forwardWhen protein X is subjected to dimethylsuberimidate and SDS electrophoresis a 50 kDa band is seen; when the protein X is subjected to SDS electrophoresis without DMS, two bands of 20 kDa and 10 kDa are seen. How many different oligomeric structures of the native protein X are possible, being consistent with these observed results? one two three none of the above which is the answrr? and explainarrow_forwardAfter staining an SDS-PAGE gel with Coomassie Blue G-250, the protein bands are visualized by de-staining the gel in a Coomassie Blue G-250 de-staining solution. This solution is made up of 10% acetic acid, 50% methanol, and 40% distilled water. How much of each of these components do you need to prepare 5 liters of Coomassie Blue G-250 de-staining solution?arrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 BiochemistryBiochemistryISBN:9781319114671Author:Lubert Stryer, Jeremy M. Berg, John L. Tymoczko, Gregory J. Gatto Jr.Publisher:W. H. Freeman
BiochemistryBiochemistryISBN:9781319114671Author:Lubert Stryer, Jeremy M. Berg, John L. Tymoczko, Gregory J. Gatto Jr.Publisher:W. H. Freeman Lehninger Principles of BiochemistryBiochemistryISBN:9781464126116Author:David L. Nelson, Michael M. CoxPublisher:W. H. Freeman
Lehninger Principles of BiochemistryBiochemistryISBN:9781464126116Author:David L. Nelson, Michael M. CoxPublisher:W. H. Freeman Fundamentals of Biochemistry: Life at the Molecul...BiochemistryISBN:9781118918401Author:Donald Voet, Judith G. Voet, Charlotte W. PrattPublisher:WILEY
Fundamentals of Biochemistry: Life at the Molecul...BiochemistryISBN:9781118918401Author:Donald Voet, Judith G. Voet, Charlotte W. PrattPublisher:WILEY BiochemistryBiochemistryISBN:9781305961135Author:Mary K. Campbell, Shawn O. Farrell, Owen M. McDougalPublisher:Cengage Learning
BiochemistryBiochemistryISBN:9781305961135Author:Mary K. Campbell, Shawn O. Farrell, Owen M. McDougalPublisher:Cengage Learning BiochemistryBiochemistryISBN:9781305577206Author:Reginald H. Garrett, Charles M. GrishamPublisher:Cengage Learning
BiochemistryBiochemistryISBN:9781305577206Author:Reginald H. Garrett, Charles M. GrishamPublisher:Cengage Learning Fundamentals of General, Organic, and Biological ...BiochemistryISBN:9780134015187Author:John E. McMurry, David S. Ballantine, Carl A. Hoeger, Virginia E. PetersonPublisher:PEARSON
Fundamentals of General, Organic, and Biological ...BiochemistryISBN:9780134015187Author:John E. McMurry, David S. Ballantine, Carl A. Hoeger, Virginia E. PetersonPublisher:PEARSON

Biochemistry
Biochemistry
ISBN:9781319114671
Author:Lubert Stryer, Jeremy M. Berg, John L. Tymoczko, Gregory J. Gatto Jr.
Publisher:W. H. Freeman

Lehninger Principles of Biochemistry
Biochemistry
ISBN:9781464126116
Author:David L. Nelson, Michael M. Cox
Publisher:W. H. Freeman

Fundamentals of Biochemistry: Life at the Molecul...
Biochemistry
ISBN:9781118918401
Author:Donald Voet, Judith G. Voet, Charlotte W. Pratt
Publisher:WILEY

Biochemistry
Biochemistry
ISBN:9781305961135
Author:Mary K. Campbell, Shawn O. Farrell, Owen M. McDougal
Publisher:Cengage Learning

Biochemistry
Biochemistry
ISBN:9781305577206
Author:Reginald H. Garrett, Charles M. Grisham
Publisher:Cengage Learning

Fundamentals of General, Organic, and Biological ...
Biochemistry
ISBN:9780134015187
Author:John E. McMurry, David S. Ballantine, Carl A. Hoeger, Virginia E. Peterson
Publisher:PEARSON
Biomolecules - Protein - Amino acids; Author: Tutorials Point (India) Ltd.;https://www.youtube.com/watch?v=ySNVPDHJ0ek;License: Standard YouTube License, CC-BY