
Organic Chemistry
9th Edition
ISBN: 9781305080485
Author: John E. McMurry
Publisher: Cengage Learning
expand_more
expand_more
format_list_bulleted
Concept explainers
Textbook Question
Chapter 6.4, Problem 5P
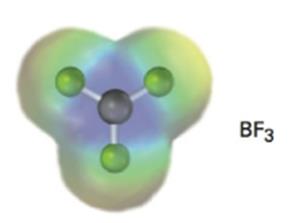
An electrostatic potential map of boron trifluoride is shown. Is BF3 likely to be a nucleophile or an electrophile? Draw a Lewis structure for BF3, and explain your answer.
Expert Solution & Answer
Trending nowThis is a popular solution!

Students have asked these similar questions
In cells, vitamin C exists largely as its conjugate base X. X is an antioxidant because radicals formed in oxidation processes abstract the labeled H atom, forming a new radical that halts oxidation. Draw the structure of the radical formed by H abstraction, and explain why this H atom is most easily removed.
In an electrophilic aromatic substitution, which group is more activating: -COH or -CH2-CH3? Explain the reason.
1. Which among these can make a molecule nucleophilic?
a.double bondsb.positive chargec. incomplete octet
2. Which among these can make a molecule electrophilic?
a.Triple bondsb.positive chargec. radicals
Chapter 6 Solutions
Organic Chemistry
Ch. 6.1 - Prob. 1PCh. 6.3 - Prob. 2PCh. 6.3 - Using curved fishhook arrows, propose a mechanism...Ch. 6.4 - Prob. 4PCh. 6.4 - An electrostatic potential map of boron...Ch. 6.5 - What product would you expect from reaction of...Ch. 6.5 - Reaction of HBr with 2-methylpropene yields...Ch. 6.6 - Prob. 8PCh. 6.6 - Predict the products of the following polar...Ch. 6.7 - Which reaction is more energetically favored, one...
Ch. 6.7 - Prob. 11PCh. 6.9 - Which reaction is faster, one with ∆G‡ = +45...Ch. 6.10 - Prob. 13PCh. 6.SE - Prob. 14VCCh. 6.SE - Prob. 15VCCh. 6.SE - Prob. 16VCCh. 6.SE - Look at the following energy diagram: (a) Is...Ch. 6.SE - Look at the following energy diagram for an...Ch. 6.SE - What is the difference between a transition state...Ch. 6.SE - Prob. 20EDRMCh. 6.SE - Prob. 21EDRMCh. 6.SE - Draw an energy diagram for a two-step exergonic...Ch. 6.SE - Draw an energy diagram for a reaction with keq =...Ch. 6.SE - The addition of water to ethylene to yield ethanol...Ch. 6.SE - When isopropylidenecyclohexane is treated with...Ch. 6.SE - Prob. 26EDRMCh. 6.SE - Draw the electron-pushing mechanism for each...Ch. 6.SE - Draw the complete mechanism for each polar...Ch. 6.SE - Prob. 29EDRMCh. 6.SE - Identify the functional groups in the following...Ch. 6.SE - Identify the following reactions as additions,...Ch. 6.SE - Identify the likely electrophilic and nucleophilic...Ch. 6.SE - For each reaction below identify the electrophile...Ch. 6.SE - Prob. 34APCh. 6.SE - Follow the flow of electrons indicated by the...Ch. 6.SE - Prob. 36APCh. 6.SE - Prob. 37APCh. 6.SE - Despite the limitations of radical chlorination of...Ch. 6.SE - Prob. 39APCh. 6.SE - Answer question 6-39 taking all stereoisomers into...Ch. 6.SE - Prob. 41APCh. 6.SE - Prob. 42APCh. 6.SE - Prob. 43APCh. 6.SE - The reaction of hydroxide ion with chloromethane...Ch. 6.SE - Prob. 45APCh. 6.SE - Ammonia reacts with acetyl chloride (CH3COCl) to...Ch. 6.SE - The naturally occurring molecule α-terpineol is...Ch. 6.SE - Prob. 48APCh. 6.SE - Prob. 49APCh. 6.SE - Draw the structures of the two carbocation...
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- Is this a base or a nucleophilearrow_forwardIdentify the electrophile and the nucleophile in each of the following reaction steps. Then draw curved arrows to illustrate the bond-making and bond-breaking processes.arrow_forwardIf the reactant provided is neutral what can be done in basic conditions to produce a nucleophile. Provide an example of an SN2 reaction where this occurs.arrow_forward
- In an electrophilic aromatic substitution, which group is more activating: -OH or -CH2-CH3? Explain the reason.arrow_forwardWhy is benzene a weak nucleophile?arrow_forwardWhich of the following species is likely to be an electrophile and which is nucleophile a) Hcl b) CH3NH2 C) CH3SH D)CH3CHOarrow_forward
- Pleas explain how this process occurs. Identify SN1, SN2, E2, E1, nucleophiles and electrophiles.arrow_forwardIdentify the appropriate nucleophile and alkyl halide substrate to prepare this ether: Structure of the alkyl halide substrate? Structure of the nucleophile?arrow_forwardWhile CH3OH is the solvent in the reaction, and a nucleophilic one, why doesn’t CH3OH act as the nucleophile in this reaction?arrow_forward
- Explain why quinuclidine is a much more reactive nucleophile than triethylamine, even though both compounds have N atoms surrounded by three R groups.arrow_forwardWhich factors would favor an SN2 reaction? Choose one or more: A. a strong nucleophile B. a good leaving group C. a high concentration of nucleophilearrow_forwardDefine Electrophilic Aromatic Substitution ?arrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning

Organic Chemistry: A Guided Inquiry
Chemistry
ISBN:9780618974122
Author:Andrei Straumanis
Publisher:Cengage Learning
General Chemistry | Acids & Bases; Author: Ninja Nerd;https://www.youtube.com/watch?v=AOr_5tbgfQ0;License: Standard YouTube License, CC-BY