
EBK EXPERIMENTAL ORGANIC CHEMISTRY: A M
6th Edition
ISBN: 9781305687875
Author: Gilbert
Publisher: CENGAGE LEARNING - CONSIGNMENT
expand_more
expand_more
format_list_bulleted
Question
Chapter 7.4, Problem 12E
Interpretation Introduction
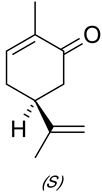
Interpretation: The GLC analysis of the oil needs to be explained which supports that there is a significant amount of a carbonyl-containing contaminant of carvone in the dill seed oil. It needs to be explained that how this proposal can be supported experimentally.
Concept Introduction: Carvone is known to be a natural product belonging to the class of terpenoids. It contains a
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
Which compound(s) in spearmint oil do you expect will be visible on the TLC plate under UV irradiation? Explain why.
Compounds:R-carvone, R-limonene and (1R,2R,4R)-dihydrocarveol
Cis-3-hexene and trans-3-hexene can be separated and analyzed using GC without
injecting an equimolar standard to a high accuracy. Explain why that is the case for these
two compounds while we had to inject an equimolar solution for our experiment
What is the order in which the following compounds would he eluted from an HPLC column containing a reversed -phase packing?(a) benzene, diethyl ether, n-hexane(b) acetone, dichloroethane, acetamide
Chapter 7 Solutions
EBK EXPERIMENTAL ORGANIC CHEMISTRY: A M
Ch. 7.2 - Prob. 1ECh. 7.2 - Prob. 2ECh. 7.2 - Prob. 3ECh. 7.2 - Prob. 4ECh. 7.2 - Prob. 5ECh. 7.2 - Prob. 6ECh. 7.2 - Prob. 7ECh. 7.2 - Prob. 8ECh. 7.2 - Prob. 9ECh. 7.2 - Prob. 10E
Ch. 7.3 - Prob. 1ECh. 7.3 - Prob. 2ECh. 7.3 - Prob. 3ECh. 7.3 - Prob. 4ECh. 7.3 - Prob. 5ECh. 7.3 - Prob. 6ECh. 7.3 - Prob. 7ECh. 7.3 - Prob. 8ECh. 7.4 - Prob. 1ECh. 7.4 - Prob. 2ECh. 7.4 - Prob. 3ECh. 7.4 - Prob. 4ECh. 7.4 - Prob. 5ECh. 7.4 - Prob. 6ECh. 7.4 - Prob. 7ECh. 7.4 - Prob. 8ECh. 7.4 - Prob. 9ECh. 7.4 - Prob. 10ECh. 7.4 - Prob. 11ECh. 7.4 - Prob. 12ECh. 7.4 - Prob. 13ECh. 7.6 - Prob. 1ECh. 7.6 - Prob. 2ECh. 7.6 - Prob. 3ECh. 7.6 - Prob. 4ECh. 7.6 - Prob. 5ECh. 7.6 - Prob. 6ECh. 7.6 - Prob. 7ECh. 7.6 - Prob. 8ECh. 7.6 - Prob. 9ECh. 7.6 - Prob. 10E
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- Complete the following table. In the second column, assign each set of molecules as constitutional isomers, conformational isomers, enantiomers, diastereomers, or identical. For the third column, based on that analysis, indicate for each set of molecules if they are separable or inseparable by SiO2 chromatography.arrow_forwardSince there are some inherent issues with polarimetric measurements of enantiomeric purity, suggest another technique that could be used to confirm the percent enantiomer excess.arrow_forwardThis is an illustration of TLC plates. The compounds used were syn-azobenzene (A) and anti-azobenzene (B). The difference between the two plates are that the first was placed in 15% methylene chloride in pet. ether and the second was placed in 30% methylene chloride in pet. ether. I calculated Rf values to be 0.47 mm for syn-azobenzene and 0.44 mm for anti-azobenzene in 15% while syn-azobenze had a Rf of 0.28 mm and 0.16 mm. Which solvent is best at separating and how does Rf help identify which isomer is which? I read that the anti is supposed to have a higher Rf but that is not the case here so I want to know if the syn is supposed to have the higher Rf and the anti is supposed to have the lower one.arrow_forward
- Greener synthesis of Bupropion Hydrochloride What spot on TLC would be higher for an eluent system of 1:1 acetone: hexane, when bupropion hydrochloride is spotted against m-chloropropiophenone?arrow_forwardThe literature value for the melting range of a pure compound is 113-115o C. What is the expected experimental melting range of that recrystallized compound immediately after being isolated from the Buchner funnel before drying completely? Explain.arrow_forwardExplain the elution sequence (GLC) of: mesitylene<1-phenylethanol<acetophenone.arrow_forward
- A neutral compound has a partition coefficient of 5 5 between ether and water. What percentage of the compound would be extracted from 10 mL of water if 10 mL of ether were used to extract the compound?arrow_forwardThere have been cases where wine has been mistakenly adulterated with ethylene glycol (,2-ethanediol, bp760 97.6 ˚C). As a forensic chemist, suggest how would you use GLC to prove conclusively that ethylene glycol was actually present?arrow_forwardWhat does an IR spectrometer measure? What gives rise to the spectral features? Why is Tm (melting temperature) positive for some DSC plots and negative for others? What does it tell us about the type of polymer being analyzed?arrow_forward
- When free in solution (i.e., not bound to opsin), both all-trans and 11-cis retinal have absorption maxima of about 370-380 nm. a. How many electrons can be counted in the conjugated pi-system?b. Using l = 370 nm and the 1D particle in a box model, estimate the effective chain length of the retinal chromophore.arrow_forwardGive and observation and inference when the compound hexane is subjected into test for Br2 in CCl4arrow_forwardGive a detailed procedure of how you will process 5 samples of palm oil adulterated with Sudan IV dye before analysing it with Raman spectroscopy. Name and state the functions reagents used in the sample processing.arrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 Macroscale and Microscale Organic ExperimentsChemistryISBN:9781305577190Author:Kenneth L. Williamson, Katherine M. MastersPublisher:Brooks Cole
Macroscale and Microscale Organic ExperimentsChemistryISBN:9781305577190Author:Kenneth L. Williamson, Katherine M. MastersPublisher:Brooks Cole

Macroscale and Microscale Organic Experiments
Chemistry
ISBN:9781305577190
Author:Kenneth L. Williamson, Katherine M. Masters
Publisher:Brooks Cole
Thermogravimetric Analysis [ TGA ] # Thermal Analysis # Analytical Chemistry Part-11# CSIR NET/GATE; Author: Priyanka Jain;https://www.youtube.com/watch?v=p1K-Jpzylso;License: Standard YouTube License, CC-BY