
(a)
Interpretation:
The bond which can be easily broken for the conversion of compound 6 to 7 and 8 needs to be determined.
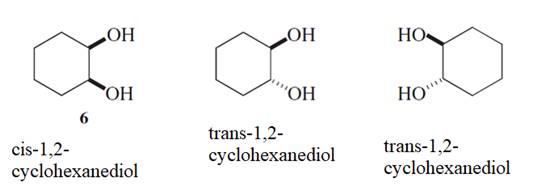
Concept Introduction :
Stereoisomers are the isomers with different spatial arrangement of the atoms, instead of order of atomic connectivity. It has the same molecular formula and the similar connectivity except for the procedure within 2D or 3D space. Like cis- and trans-but-2-ene both have two CH3 groups 2-H and a C=C but connectivity is different in the space.
(b)
Interpretation:
The value of
Concept Introduction :
Stereoisomers are the isomers with different spatial arrangement of the atoms, instead of order of atomic connectivity. It has the same molecular formula and the similar connectivity except for the procedure within 2D or 3D space. Like cis- and trans-but-2-ene both have two CH3 groups 2-H and a C=C but connectivity is different in the space.
Want to see the full answer?
Check out a sample textbook solution
Chapter 7 Solutions
EBK EXPERIMENTAL ORGANIC CHEMISTRY: A M
- The pKa of cyclopentane is > 60, which is about what is expected for a hydrogen that is bonded to an sp3 carbon. Explain why cyclopentadiene is a much stronger acid (pKa of 15) even though it too involves the loss of a proton from an sp3 carbon.arrow_forwardkindly write a brief description of where each fragment peak originated from the parent ion. example: Formation of m/z 79 ion: [CH3CH2Br]+ ===> [79Br]^+ + CH2CH3. ( or a diagram of each bond breaking / forming could also work )arrow_forwardConsider the structures of cholesterol and ?-carotene. Which do you think will have the highest Rf value if the solvent used was acetone - hexane (20:80)? Why?arrow_forward
- Calculate the Rf values for each color component and arrange them in terms of decreasing polarity. Explain how did you come up with this sequence.arrow_forwardSolvent #1 is a 50:50 mixture of hexane and ethyl acetate and solvent #2 is 25% hexane. Which solvent mixture is more polar: the 50:50 mixture or the 25:75 mixture? Explain briefly What would happen to the Rf of the spots in solvent #2 compared to solvent #1? Why?arrow_forwarda) which bond is present in alcohols but not alkanes? b) is this band strong or weak (in terms of spectra)? c) what is different about C=O and O-H bonds compared with C=C or C≡C bonds that show weaker signals?arrow_forward
- Briefly explain based on the IR spectra how well the reaction worked.arrow_forwardCan someone help me figure out which analytical method would be the most effective for this probelm? I don't think B (melting pt) and C (infrared spectroscopy= functional groups) are the most effective.arrow_forwardThis is about Beer-Lambert’s Law and UV-vis Spectroscopy: 1. How do you make a calibration graph?2. How do you use Beer-Lambert’s Law?3. How does the solution color relate to its absorbance spectrum?arrow_forward
- QUESTION 6 EXPLAIN CAREFULLY WITH A DRAWING WHY THE PROTONS SUBSTITUTED ORTHO TO AN ACETYL GROUP NORMALLY HAVE A GREATER CHEMICAL SHIFT THAN THE OTHER PROTONS ON THE RING.arrow_forwardExplain briefly the differences in Rf values of the pigments that you have separated from spinach juice. What properties must the pigment have to travel further in the chromatogram? Consider the structures of cholesterol and ?-carotene. Which do you think will have the highest Rf value if the solvent used was acetone - hexane (20:80)? Why?arrow_forward1. a. Why would a column be ineffective if you chose an eluting solvent that was too polar? b. Why would a column be ineffective if you chose an eluting solvent that was not polar enough?arrow_forward
 Macroscale and Microscale Organic ExperimentsChemistryISBN:9781305577190Author:Kenneth L. Williamson, Katherine M. MastersPublisher:Brooks Cole
Macroscale and Microscale Organic ExperimentsChemistryISBN:9781305577190Author:Kenneth L. Williamson, Katherine M. MastersPublisher:Brooks Cole
