
EBK EXPERIMENTAL ORGANIC CHEMISTRY: A M
6th Edition
ISBN: 9781305687875
Author: Gilbert
Publisher: CENGAGE LEARNING - CONSIGNMENT
expand_more
expand_more
format_list_bulleted
Question
Chapter 7.4, Problem 7E
Interpretation Introduction
Interpretation:
The percentage of the carvone present in spearmint and caraway seed oils needs to be estimated.
Concept Introduction :
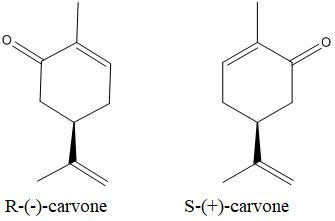
The isomers which are non-superimposable mirror image of each other are known as enantiomers.
The enatiomers of the carvone is as below:
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
Cis-3-hexene and trans-3-hexene can be separated and analyzed using GC without
injecting an equimolar standard to a high accuracy. Explain why that is the case for these
two compounds while we had to inject an equimolar solution for our experiment
Why was n-octanol chosen as a surrogate for natural organic phases? Why notanother solvent such as n-hexane, methylbenzene, trichloromethane, or diethylether?Why is the use of any organic solvent as general surrogate of a natural organicphase somewhat questionable?
Please help me solve for the epoxyproduct:
Please calculate the amount expected for the epoxyproduct.
Name
Molecula Weight
Amount Used
mmol
Equivalents
Density
Carvone
150.22
.72mL
4.792
1
.9650
H2O2
34.01
1.5mL
48.95
10.58
10.58
NaOH
40
1 mL
6
1.297
-
Methanol
32.04
8 mL
197.5
42.7
.791
Diethyl Ether
74.12
-
-
-
.706
Ethyl Acetate
88.11
-
-
-
.902
Hexanes
86.17
-
-
-
.659
Phosphomolybdic Acid
1825.25
-
-
-
1.62
Epoxyproduct
166.22
-
?
Amount Expected
-
Chapter 7 Solutions
EBK EXPERIMENTAL ORGANIC CHEMISTRY: A M
Ch. 7.2 - Prob. 1ECh. 7.2 - Prob. 2ECh. 7.2 - Prob. 3ECh. 7.2 - Prob. 4ECh. 7.2 - Prob. 5ECh. 7.2 - Prob. 6ECh. 7.2 - Prob. 7ECh. 7.2 - Prob. 8ECh. 7.2 - Prob. 9ECh. 7.2 - Prob. 10E
Ch. 7.3 - Prob. 1ECh. 7.3 - Prob. 2ECh. 7.3 - Prob. 3ECh. 7.3 - Prob. 4ECh. 7.3 - Prob. 5ECh. 7.3 - Prob. 6ECh. 7.3 - Prob. 7ECh. 7.3 - Prob. 8ECh. 7.4 - Prob. 1ECh. 7.4 - Prob. 2ECh. 7.4 - Prob. 3ECh. 7.4 - Prob. 4ECh. 7.4 - Prob. 5ECh. 7.4 - Prob. 6ECh. 7.4 - Prob. 7ECh. 7.4 - Prob. 8ECh. 7.4 - Prob. 9ECh. 7.4 - Prob. 10ECh. 7.4 - Prob. 11ECh. 7.4 - Prob. 12ECh. 7.4 - Prob. 13ECh. 7.6 - Prob. 1ECh. 7.6 - Prob. 2ECh. 7.6 - Prob. 3ECh. 7.6 - Prob. 4ECh. 7.6 - Prob. 5ECh. 7.6 - Prob. 6ECh. 7.6 - Prob. 7ECh. 7.6 - Prob. 8ECh. 7.6 - Prob. 9ECh. 7.6 - Prob. 10E
Knowledge Booster
Similar questions
- Assuming that either solvent is otherwise acceptable in a given instance, what advantages does ethanol have over 1-octanol as a crystallization solvent? Hexane over pentane? Water over methanol?arrow_forwardWe want to extract terpenoids from an aqueous sample by continuous liquid-liquid extraction. Which of the following solvents is not suitable for this purpose? Why? Propanone (acetone), dichloromethane, heptane.arrow_forwardCOMPARE the two diagrams: a. phase diagram of eutectic point between binary system of menthol and camphor and b. phase diagram of the theoretical eutectic points. (handwritten diagram) what were the factors that could have affected the results? provide recommendations on how to increase the accuracy/correctness of dataarrow_forward
- The procedure for the synthesis of menthone indicates that you should use 10 cm³ of 6M sulphuric acid. Suppose that only sulphuric acid with concentration 1.5M is available in the laboratory. What volume in cubic centimetres of this solution do you need to add to the reaction mixture to replace the solution originally specified?arrow_forwardChemistry Discuss the possible consequences of injecting the concentrated injectable suspension above through a hypodermic needle?arrow_forwardArrange the following compounds in order of decreasing ease of elution from a column of silica gel: (a) 2-octanol; (b) 1,3-dichlorobenzene; (c) tert-butylcyclo hexane; (d) benzoic acid.arrow_forward
- Give the functions of the following equipment in a vacuum distillation setup: -vacuum adapter-vacuum pump-manometer-water bath-claisen flask-capillary tube-stopcock-thermometer-inlet and outlet nozzle-condenser-receiving flask Kindly include all please, one sentence each will do. I will upvotearrow_forwardDiscuss the structure of phospholipids. Explain the dependence of freezing (gel to liquid crystal transition) temperature for bilayer phospholipid membranes on the length of the tail and the number of double bonds in the fatty acid chains of phospholipids.arrow_forwardIn the experiment borohydride reduction , using 2.0g of benzophenone, a student obtained 1.6g of filtered material. Calculate the percentage yield and comment on whether this represents the true yield of the reaction product diphenylmethanol.arrow_forward
- 1.Why are the chlorophylls less mobile than the carotenes on the TLC plate? Give athorough explanation, including a sketch of how each type of molecule would interactwith the silica stationary phase material (you can use schematic structure formulas, ratherthan draw the whole molecules, which are quite large!). For instance, would one of thosetypes of molecules create more intermolecular force interactions than the other?arrow_forward1. Why should the stopper always be removed from a separatory funnel whenever a liquid is being drained through the stopcock?2. The distribution coefficient, KD (ether/water), between ether and water for aspirin at room temperature is 3.5. What weight of aspirin would be extracted by a single extraction with 60 ml of water from a solution of 10 grams of aspirin in 100 ml of ether? Calculate the weight of aspirin which would be removed by three extractions with 20 ml portions of water.arrow_forwardDescribe in words and by means of complete chemical equations, how a mixture of 1.25 g of benzoic acid tertbutylbenzene can be separated by applying the separation of 2 components by liquid-liquid extraction.arrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Macroscale and Microscale Organic ExperimentsChemistryISBN:9781305577190Author:Kenneth L. Williamson, Katherine M. MastersPublisher:Brooks Cole
Macroscale and Microscale Organic ExperimentsChemistryISBN:9781305577190Author:Kenneth L. Williamson, Katherine M. MastersPublisher:Brooks Cole

Principles of Instrumental Analysis
Chemistry
ISBN:9781305577213
Author:Douglas A. Skoog, F. James Holler, Stanley R. Crouch
Publisher:Cengage Learning

Macroscale and Microscale Organic Experiments
Chemistry
ISBN:9781305577190
Author:Kenneth L. Williamson, Katherine M. Masters
Publisher:Brooks Cole