
EBK EXPERIMENTAL ORGANIC CHEMISTRY: A M
6th Edition
ISBN: 9781305687875
Author: Gilbert
Publisher: CENGAGE LEARNING - CONSIGNMENT
expand_more
expand_more
format_list_bulleted
Question
Chapter 7.4, Problem 8E
(a)
Interpretation Introduction
Interpretation:
The percentage of the carvone present in the dill seed oil needs to be calculated.
Concept Introduction :
The isomers which are a non-superimposable mirror images of each other are known as enantiomers.
The enatiomers of the carvone is as below:
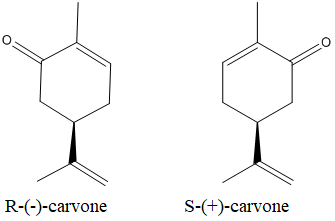
(b)
Interpretation Introduction
Interpretation:
The reason for the retention time of the carvone in the oil to not identify the type of enantiomer present in it needs to be explained.
Concept Introduction :
The isomers which arenon-superimposable mirror images of each other are known as enantiomers.
The enantiomers of the carvone are as below:
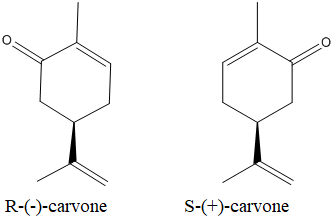
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
In glucose estimation using Benedict's Reagent, briefly explain what would happen if the water evaporates completely and is not immediately replaced and what effect this will have on the calculated titer of Benedict's reagent.
Which combination cannot be used as mobile phase in RPLC?
A. Acetonitrile-cyclopentane
B. Water-acetonitrile
C. Acetonitrile-THF
D. Water-THF
Silica gel is very polar stationary phase. Therefore, more polar molecule sticks to silica gel on the TLC plate and has higherRfvalue.
True
False
Chapter 7 Solutions
EBK EXPERIMENTAL ORGANIC CHEMISTRY: A M
Ch. 7.2 - Prob. 1ECh. 7.2 - Prob. 2ECh. 7.2 - Prob. 3ECh. 7.2 - Prob. 4ECh. 7.2 - Prob. 5ECh. 7.2 - Prob. 6ECh. 7.2 - Prob. 7ECh. 7.2 - Prob. 8ECh. 7.2 - Prob. 9ECh. 7.2 - Prob. 10E
Ch. 7.3 - Prob. 1ECh. 7.3 - Prob. 2ECh. 7.3 - Prob. 3ECh. 7.3 - Prob. 4ECh. 7.3 - Prob. 5ECh. 7.3 - Prob. 6ECh. 7.3 - Prob. 7ECh. 7.3 - Prob. 8ECh. 7.4 - Prob. 1ECh. 7.4 - Prob. 2ECh. 7.4 - Prob. 3ECh. 7.4 - Prob. 4ECh. 7.4 - Prob. 5ECh. 7.4 - Prob. 6ECh. 7.4 - Prob. 7ECh. 7.4 - Prob. 8ECh. 7.4 - Prob. 9ECh. 7.4 - Prob. 10ECh. 7.4 - Prob. 11ECh. 7.4 - Prob. 12ECh. 7.4 - Prob. 13ECh. 7.6 - Prob. 1ECh. 7.6 - Prob. 2ECh. 7.6 - Prob. 3ECh. 7.6 - Prob. 4ECh. 7.6 - Prob. 5ECh. 7.6 - Prob. 6ECh. 7.6 - Prob. 7ECh. 7.6 - Prob. 8ECh. 7.6 - Prob. 9ECh. 7.6 - Prob. 10E
Knowledge Booster
Similar questions
- Which combination cannot be used as mobile phase in RPLC? A. Water-acetonitrile B. Acetonitrile-cyclopentane C. water-THF (tetrahydrofuran) D. Acetonitrile-THF (tetrahydrofuran)arrow_forwardWhen a pure sample of tert-butyl bromide is analyzed by gas chromatography, two components are usually observed. One of them is tert-butyl bromide and the other one is a decomposition product. As the temperature of the injector is increased, the amount of the decomposition product increases and the amount of tert-butyl bromide decreases.a. What is the structure of the decomposition product?b. Why does the amount of decomposition increase with increasing temperature?c. Why does tert-butyl bromide decompose much more easily than tert-butyl chloride?arrow_forwardWhich is more polar, the water solvent or the chromatogrphy paper?arrow_forward
- what is more polar, pyrrole or benzaldehyde? I need to know for a TLC test/ which one should be further up?arrow_forwardWhich compound(s) in spearmint oil do you expect will be visible on the TLC plate under UV irradiation? Explain why. Compounds:R-carvone, R-limonene and (1R,2R,4R)-dihydrocarveolarrow_forwardIn reversed phase partition chromatography Select one: a. the least polar compound elutes first b. the smallest molecular weight compound elutes first c. the most polar compound elutes first d. the largest molecular weight compound elutes first e. the lowest boiling compound elutes first f. the highest boiling compound elutes firstarrow_forward
- Based on the resulting chromatograms after visualization with iodine vapor, which between the solvent systems WHITE and MIX resulted in a more effective separation of the pigments of the indigo extract? Explain your answer in terms of the solvent that did not effectively separate the components of the root extract.arrow_forwardDiethylacetal can be detected by gas chromatography. How does this impurity arise in fermentation?arrow_forwarddo you think that a dye with a high retention factor is more soluble or less souluble in the solvent than a dye with a lower retention factor? why?arrow_forward
- Explain the elution sequence (GLC) of: mesitylene<1-phenylethanol<acetophenone.arrow_forwardSpinach chromatography lab: provide a short description of the following processes: a) Spinach treatment b) Preparing a chromatography column c) Collecting the two major specifies (β-carotene and Chlorophyll A) d) Spectra measurementarrow_forwardWhich of the following carotenoid structures below would you expect to have the lowest RF in a chromatography experiment using an ether-petroleum ether mobile phase? Why? Lycopene: β-carotene: Vitamin Aarrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 EBK A SMALL SCALE APPROACH TO ORGANIC LChemistryISBN:9781305446021Author:LampmanPublisher:CENGAGE LEARNING - CONSIGNMENT
EBK A SMALL SCALE APPROACH TO ORGANIC LChemistryISBN:9781305446021Author:LampmanPublisher:CENGAGE LEARNING - CONSIGNMENT

EBK A SMALL SCALE APPROACH TO ORGANIC L
Chemistry
ISBN:9781305446021
Author:Lampman
Publisher:CENGAGE LEARNING - CONSIGNMENT