
Concept explainers
a)
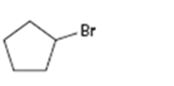
Interpretation:
The alkene that will give 1-bromocyclopentane in an addition reaction is to be identified.
Concept introduction:
In addition reactions small molecules like HCl, H2O, HBr etc. add to the double bond in an alkene to yield a single product. The addition to unsymmetrical
To state:
Which alkene will give 1-bromocyclopentane as product in an addition reaction.
b)
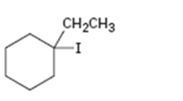
Interpretation:
The alkene that will give 1-ethyl-1-iodocyclopentane in an addition reaction is to be identified.
Concept introduction:
In addition reactions small molecules like HCl, H2O, HBr etc. add to the double bond in an alkene to yield a single product. The addition to unsymmetrical alkenes will follow Markovnikov’s rule which states that “in the addition of HX to an alkene, the H adds to the carbon with fewer alkyl substituents and X adds to the carbon with more alkyl substituents”.
To state:
Which alkene will give 1-ethyl-1-iodocyclohexane as product in an addition reaction.
c)
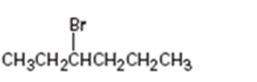
Interpretation:
The alkene that will give 3-bromohexane in an addition reaction is to be identified.
Concept introduction:
In addition reactions small molecules like HCl, H2O, HBr etc. add to the double bond in an alkene to yield a single product. The addition to unsymmetrical alkenes will follow Markovnikov’s rule which states that “in the addition of HX to an alkene, the H adds to the carbon with fewer alkyl substituents and X adds to the carbon with more alkyl substituents”.
To state:
Which alkene will give 3-bromohexane as product in an addition reaction.
d)
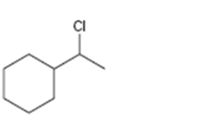
Interpretation:
The alkene that will give 1-chloroethylcyclopentane in an addition reaction is to be identified.
Concept introduction:
In addition reactions small molecules like HCl, H2O, HBr etc. add to the double bond in an alkene to yield a single product. The addition to unsymmetrical alkenes will follow Markovnikov’s rule which states that “in the addition of HX to an alkene, the H adds to the carbon with fewer alkyl substituents and X adds to the carbon with more alkyl substituents”.
To state:
Which alkene will give 1-chloroethylcyclohexane as product in an addition reaction.
Trending nowThis is a popular solution!

Chapter 7 Solutions
Organic Chemistry
- How could the following compounds be prepared using an alkene as one of the starting materials?arrow_forwardHow would you prepare the following alkyl halides, from alkenes? Write the equation for each reaction indicating substrates, reagents, and conditions.arrow_forwardStarting with cyclohexane, how could the following compounds be prepared?arrow_forward
- Provide the major product for the reaction of the following starting material with H2O, H+. If more than one product is formed, list the major product first. If no reaction, draw the starting material.arrow_forwardWhat are the reagents of the following reactions?arrow_forwardHow could the following compound be prepared using an alkene as one of the starting materials?arrow_forward
- What alkene and reagent should be used to synthesize the following alkyl halide?arrow_forwardUsing bromocyclohexane as a starting material, how could you synthesize the following compounds?arrow_forwardUsing any alkene and any other reagents, how would you prepare the following compounds?arrow_forward

