
a)
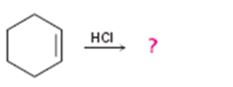
Interpretation:
The product of the reaction shown is to be predicted.
Concept introduction:
In addition reactions small molecules like HCl, H2O, HBr etc. add to the double bond in an alkene to yield a single product. The addition to unsymmetrical
To predict:
The product in the reaction shown.
b)
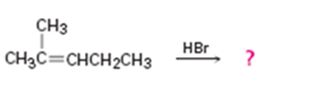
Interpretation:
The product in the reaction shown is to be predicted.
Concept introduction:
In addition reactions small molecules like HCl, H2O, HBr etc. add to the double bond in an alkene to yield a single product. The addition to unsymmetrical alkenes will follow Markovnikov’s rule which states that “in the addition of HX to an alkene, the H adds to the carbon with fewer alkyl substituents and X adds to the carbon with more alkyl substituents”.
To predict:
The product in the reaction shown.
c)
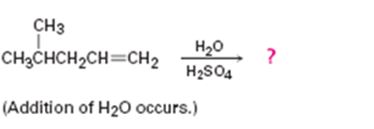
Interpretation:
The product in the reaction is to be predicted.
Concept introduction:
In addition reactions small molecules like HCl, H2O, HBr etc. add to the double bond in an alkene to yield a single product. The addition to unsymmetrical alkenes will follow Markovnikov’s rule which states that “in the addition of HX to an alkene, the H adds to the carbon with fewer alkyl substituents and X adds to the carbon with more alkyl substituents”.
To predict:
The product in the reaction shown.
d)
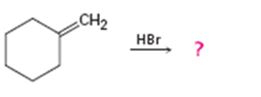
Interpretation:
The product in the reaction is to be predicted.
Concept introduction:
In addition reactions small molecules like HCl, H2O, HBr etc. add to the double bond in an alkene to yield a single product. The addition to unsymmetrical alkenes will follow Markovnikov’s rule which states that “in the addition of HX to an alkene, the H adds to the carbon with fewer alkyl substituents and X adds to the carbon with more alkyl substituents”.
To predict:
The product in the reaction shown.
Trending nowThis is a popular solution!

Chapter 7 Solutions
Organic Chemistry
 Principles of Modern ChemistryChemistryISBN:9781305079113Author:David W. Oxtoby, H. Pat Gillis, Laurie J. ButlerPublisher:Cengage Learning
Principles of Modern ChemistryChemistryISBN:9781305079113Author:David W. Oxtoby, H. Pat Gillis, Laurie J. ButlerPublisher:Cengage Learning
