
Concept explainers
a)
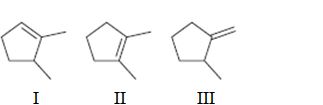
Interpretation:
To rank the double bond in the compounds given in terms of increasing stability.
Concept introduction:
The stability of a double bond can be determined by the number of hydrogens on adjacent carbons attached to both carbons in the double bond. (H’s on the α carbon). More the number of such hydrogens, the more hyperconjugation occur and more stable the
b)
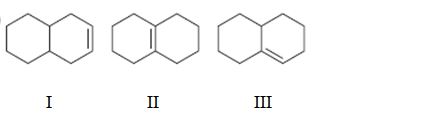
Interpretation:
To rank the double bond in the compounds given in terms of increasing stability.
Concept introduction:
The stability of a double bond can be determined by the number of hydrogens on adjacent carbons attached to both carbons in the double bond.(H’s on the α carbon). More the number of such hydrogens, the more hyperconjugation occur and more stable the alkene.
c)
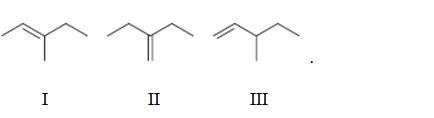
Interpretation:
To rank the double bond in the compounds given in terms of increasing stability.
Concept introduction:
The stability of a double bond can be determined by the number of hydrogens on adjacent carbons attached to both carbons in the double bond.(H’s on the α carbon). More the number of such hydrogens, the more hyperconjugation occur and more stable the alkene.
Trending nowThis is a popular solution!

Chapter 7 Solutions
Organic Chemistry
- Answer the following questions about erlotinib and terbinafine. Erlotinib,sold under the trade name Tarceva, was introduced in 2004 for thetreatment of lung cancer. Terbinafine is an antifungal medication used totreat ringworm and fungal nail infections. Rank the labeled bonds in terbinafine in order of increasing bondstrength.arrow_forward(a) What orbitals are used to form the indicated bonds in α-sinensal?(b) In what type of orbitals do the lone pairs on O reside?arrow_forwardRank the indicated C–H bonds according to increasing strength.arrow_forward
- Draw all possible structural isomers (constitutional and cis-trans) in bond-line presentation. a) C3H7N (non cyclic compounds only) (should be 8) b) C2H4F2 (not many possibilities) c)F2CCH2 (pay attention that the conectivity in this compound is already defined)arrow_forwardIdentify the type of configurational isomerism that exist for each double bond. E or Zarrow_forwardExplain why alkene A is more stable than alkene B, even though Bcontains more carbon atoms bonded to the double bond. Would youexpect C to be more or less stable than A and B?arrow_forward
- The most stable MO of 1,3,5-hexatriene and the most stable MO of benzene Which compound is more stable? Why?arrow_forwardRank the carbocation compounds below from most stable to least stable.arrow_forwardRank the polarity of the indicated bonds in structures I-III from most polar to least polar.arrow_forward
- Arrange the following molecules from its increasing stability.arrow_forwardDraw the following molecule in its two most stable forms. Provide an explanation to why each form is or isn't stable.arrow_forwardDraw the structures of the free radicals in the compound, then arrange them in a decreasing order of stability.arrow_forward
 Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning

