
Interpretation:
Series of chain initiation, propagation and termination steps for the reaction and estimate its heat of reaction has to be proposed.
Concept introduction:
Halogenation of
Radical chain reaction:
Initiation reaction: 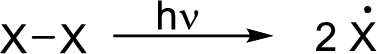
Chain propagation: 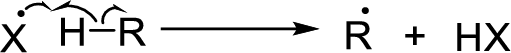
Chain termination:
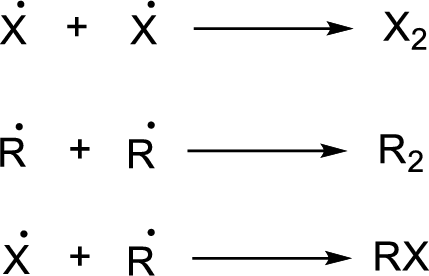
Heat of reaction for a radical reaction can be identified form bond dissociation enthalpy.
It is a change in enthalpy of a homolysis reaction at absolute zero where a molecule is broken down into two free radicals.
Trending nowThis is a popular solution!

Chapter 8 Solutions
Organic Chemistry
- One of the two diastereomers shown below reacts with potassium tertbutoxide significantly faster than then the other. Which one is it? Provide an explanation and predict the product of the fast reaction.arrow_forwardWhich of the following undergoes SN1 reaction at the fastest rate? Explain.arrow_forwardi need to propose a synthesis pathway and reagents for the preparation of the following compound. (+/-) indicates a racemic connection. in a) im looking for a reaction that can give an anti-markinov product by addition, it could be something with boron. in b) its something with addition to conjugated dienes.arrow_forward
- Provide a reasonable stepwise mechanism for the following transformation. Show the structure of any intermediates and use the arrow formalism to account for electron flow. Draw the structure of the regioisomer that might form and suggest a reason why it would not be produced.arrow_forwardPredict the major products of the following reaction. If it is possible, write all stereoisomers.arrow_forwardWhat are the possible constitutional isomers and rank by SN2 reaction.arrow_forward
- Could you show the structural formulas of the compounds A-C, whitch of these compounds is the most likely to form if the reaction occures via a chair conformation intermediate and why?.arrow_forward(d) Illustrate and briefly explain the mechanism of reaction II.arrow_forwardWhat happens to the stereochemistry during and SN2 reaction? Why? Provide a reaction to illustrate this.arrow_forward
- Provide the mechanism of the redica reaction below (b)arrow_forwardCan you please predict the MAJOR product or provide the reagents/conditions for the following reactions? Please indicate racemic if it is and if there is no reaction, please specify that as well. ?arrow_forwardThe diene below when heated,will automatically cyclize into the cyclobutene shown . Explain how this reaction is possible accounting for the stereochemical outcomesarrow_forward
 Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning EBK A SMALL SCALE APPROACH TO ORGANIC LChemistryISBN:9781305446021Author:LampmanPublisher:CENGAGE LEARNING - CONSIGNMENT
EBK A SMALL SCALE APPROACH TO ORGANIC LChemistryISBN:9781305446021Author:LampmanPublisher:CENGAGE LEARNING - CONSIGNMENT


