Part 1: Synthesis of Ligand. In this experiment you prepare the chiral ligand 4 by first converting the salt 2 to its free amine 2' then condensing it with aldehyde 3a to give bis-imine 4. t-Bu t-Bu „NH3 O2C HO NH2 OH i) K2CO3 ethanol, reflux "NH3 O2C "., “NH2 OH OH t-Bu t-Bu 2 2' t-Bu t-Bu За 1. NMR analysis of compound 4 is not performed. By comparing the structures of free amine 2' and aldehyde 3a with the bis-imine 4 indicate which two signals in the 'H-NMR (of 4) would be most useful to confirm its formation? Ensure you explain your answer and use appropriately labelled diagrams to illustrate. (Hint. Which chemical environments change the most when forming 4 from 2' and 3a)
Part 1: Synthesis of Ligand. In this experiment you prepare the chiral ligand 4 by first converting the salt 2 to its free amine 2' then condensing it with aldehyde 3a to give bis-imine 4. t-Bu t-Bu „NH3 O2C HO NH2 OH i) K2CO3 ethanol, reflux "NH3 O2C "., “NH2 OH OH t-Bu t-Bu 2 2' t-Bu t-Bu За 1. NMR analysis of compound 4 is not performed. By comparing the structures of free amine 2' and aldehyde 3a with the bis-imine 4 indicate which two signals in the 'H-NMR (of 4) would be most useful to confirm its formation? Ensure you explain your answer and use appropriately labelled diagrams to illustrate. (Hint. Which chemical environments change the most when forming 4 from 2' and 3a)
Principles of Instrumental Analysis
7th Edition
ISBN:9781305577213
Author:Douglas A. Skoog, F. James Holler, Stanley R. Crouch
Publisher:Douglas A. Skoog, F. James Holler, Stanley R. Crouch
Chapter14: Applications Of Ultraviolet-visible Molecular Absorption Spectrometry
Section: Chapter Questions
Problem 14.7QAP: A 3.03-g petroleum specimen was decomposed by wet ashing and subsequently diluted to 500 mL in a...
Related questions
Question
Need help with this question. Thank you :)
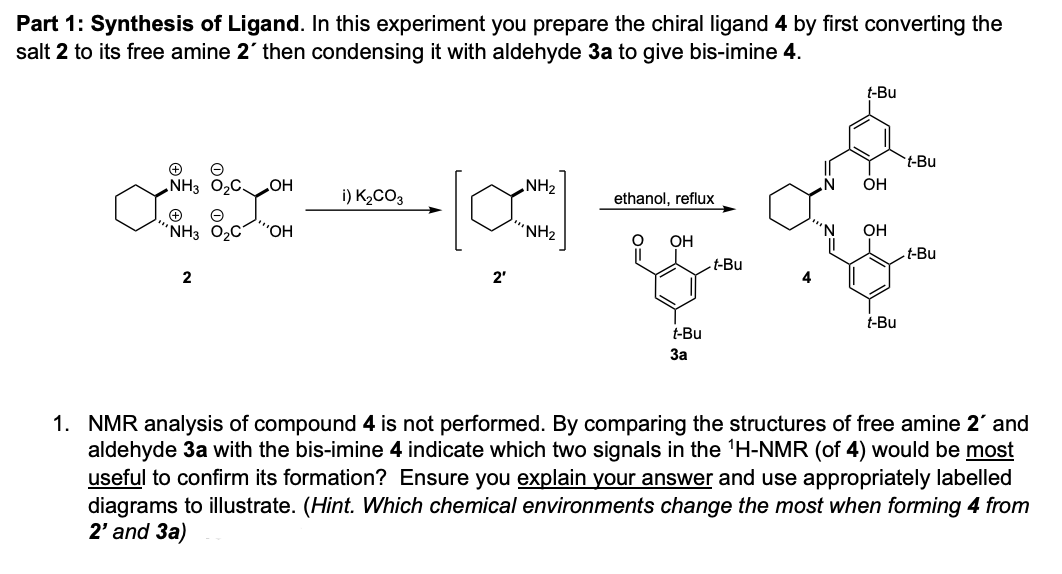
Transcribed Image Text:Part 1: Synthesis of Ligand. In this experiment you prepare the chiral ligand 4 by first converting the
salt 2 to its free amine 2' then condensing it with aldehyde 3a to give bis-imine 4.
t-Bu
t-Bu
„NH3 O2C
HO
NH2
OH
i) K2CO3
ethanol, reflux
"NH3 O2C
".,
“NH2
OH
OH
t-Bu
t-Bu
2
2'
t-Bu
t-Bu
За
1. NMR analysis of compound 4 is not performed. By comparing the structures of free amine 2' and
aldehyde 3a with the bis-imine 4 indicate which two signals in the 'H-NMR (of 4) would be most
useful to confirm its formation? Ensure you explain your answer and use appropriately labelled
diagrams to illustrate. (Hint. Which chemical environments change the most when forming 4 from
2' and 3a)
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
Step by step
Solved in 4 steps with 4 images

Recommended textbooks for you

Principles of Instrumental Analysis
Chemistry
ISBN:
9781305577213
Author:
Douglas A. Skoog, F. James Holler, Stanley R. Crouch
Publisher:
Cengage Learning


Principles of Instrumental Analysis
Chemistry
ISBN:
9781305577213
Author:
Douglas A. Skoog, F. James Holler, Stanley R. Crouch
Publisher:
Cengage Learning
