
Interpretation:Hybridization scheme that gives best description of the carbons marked by asterisks and types of orbitals are used in these bonds should be identified. Whether the bond is expected to be stronger or weaker than an ordinary
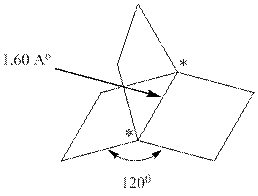
Concept introduction:
In accordance with VSEPR model, if four bonds are present around the central atom the hybridization associated is
Similarly, if three bonds are present around the central atom the hybridization associated is
The table that relates the hybridization with bond angles is as follows:
Want to see the full answer?
Check out a sample textbook solution
Chapter 1 Solutions
Organic Chemistry: Structure and Function
- There are two compounds with the molecular formula HN3. One is called hydrogen azide; the other is cyclotriazene. (a) Write the Lewis structure for each compound. (b) Designate the hybridization of each nitrogen in hydrogen azide. (c) What is the hybridization of each nitrogen in cyclotriazene? (d) How many sigma bonds are in hydrogen azide? In cyclotriazene? (e) How many pi bonds are in hydrogen azide? In cyclotriazene? (f) Give approximate values for the N-to-N-to-N bond angles in each molecule.arrow_forward9.13 (a) An AB2 molecule is linear. How many nonbonding electronpairs are around the A atom from this information?(b) How many nonbonding electrons surround the Xe inXeF2? (c) Is XeF2 linear?arrow_forwardThe following molecular orbital diagram belongs to a molecule of the type XH2 (where X is an unidentified 2nd row element). Do you think the molecule is bent or linear? Explain your reasoning. Make approximate sketches of the bonding and non-bonding molecular orbitals. If each bond in the molecule has a bond order of 1, identify X and explain your reasoning based on the MO diagram.arrow_forward
- 2. What’s the bond order of the diatomic molecule Ne2? Is your result consistent with the fact that Ne is a monatomic molecule in nature? Explain. 3. How many unpaired electrons doe the ion have? Is your result consistent with the fact that is attracted by magnet? Explainarrow_forward(a) Dihydrogen cation H2+ is commonly found in interstellar space. It can be generated by ionization of dihydrogen molecule H2. What electromagnetic radiation can be used to generate the H2+ cation? (b) Construct an energy level diagram for the molecular orbitals of the H2+ cation. What is the bond order of the HH bond in the H2+ cation? (c) Suggest a spectroscopic method for differentiation of H2 gas and the H2+ cation. Explain your answers.arrow_forwardFor the B 2H 6 molecule i) Using projection operators, derive a set of SALCs using the bridging H 1s orbitals. ii) Using projection operators, derive a set of SALCs using the s- and p- orbitals of the B atoms. iii) Using the SALCs derived in (i) and (ii), draw a molecular orbital diagram for the interaction between the B atoms and the bridging H atoms. iv) Fill in the molecular orbital diagram from (iii), remembering that 8 of the available electrons will be used in the bonds between the B atoms and the terminal H atoms – those will not show up in this diagram.arrow_forward
- Let us construct the molecular orbital diagram of ethylene (in pieces). a. First, construct the MO diagram of linear carbene (CH2). Draw pictures of all 6 orbitals b. Now bend the carbene to a bond angle of about 120°. How does this change your MO diagram? Draw pictures of all 6 orbitals. c. Now bring two of these carbene molecules together to make ethylene. Draw pictures of all 12 orbitals.arrow_forwarda) Consider a molecule of acetone cyanohydrin, shown below. What is the hybridization and molecular geometry at CA, CB, and O? (b) For Carbon B (CB) and nitrogen (N), draw orbital energy diagrams showing the orbitals involved in bonding after hybridization. Be sure to fill the orbitals appropriately with valence electrons. c) In your diagrams in part (b), which orbitals are involved in sigma and pi bonding? Orbitals Involved in Sigma Bonding Orbitals Involved in Pi Bonding 2p sp sp2 sp3 2p sp sp2 sp3 (d) In a sample containing many molecules of acetone cyanohydrin (and no other substances), what type(s) of intermolecular forces could be present? Choose all that apply. Covalent Bonds Hydrogen Bonds Dipole-Dipole Forces London Dispersion Forcesarrow_forwarda) Use the valence electron molecular orbital diagram for O2 to propose ground state O2 ions that satisfy the following conditions:(i) An O2 ion that is paramagnetic and that has a non-integer bond order.(li) An O2 ion that is paramagnetic and that has an integer bond order.(ili) An O2 ion that is diamagnetic and that has a bond order of 3.Clearly show the charge on your ion, label all valence molecular orbitals, and show the corresponding electrons in their orbitals. Full calculations for bond orders must be included.b) Using only the valence electron molecular orbital diagram for O2, is aground state diamagnetic ion with a bond order of 2 possible? Write one or twosentences to explain why or why not.arrow_forward
- 2) For each pair of molecules, generate the relevant MO diagram and predict which of the twowill have the highest bond order. (Be sure to include all valenceorbitals, and show the relative positions of the orbitals for each constituent atom.)a) HF vs HF-b) NP vs NP+c) CS vs CS+arrow_forwardThe picture below represents a molecular orbital from 1, 3 – butadiene. How many nodes does this orbital have? Is this a bonding or anti-bonding molecular orbital?arrow_forward30W.) When carbon undergoes sp³ hybridization, one sp³ hybridized orbital is formedX.) Carbon dioxide is formed when a carbon fuel undergone complete combustionY.) According to the Aufbau's principle, an orbital cannot have more than two electronsZ.) Phosphorus is from Group VA, therefore, it has 5 energy levels (5 "rings")A.) If all 4 statements are trueB.) If 3 of the 4 statements are trueC.) If 2 of the 4 statements are trueD.) If only 1 of the 4 statements is trueE.) If none of the 4 statements is truearrow_forward
 Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning
Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning General Chemistry - Standalone book (MindTap Cour...ChemistryISBN:9781305580343Author:Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; DarrellPublisher:Cengage Learning
General Chemistry - Standalone book (MindTap Cour...ChemistryISBN:9781305580343Author:Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; DarrellPublisher:Cengage Learning

