
Concept explainers
(a)
Interpretation:The geometry of indicated atom in the given molecule with the help of hybridization needs to be determined.
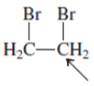
Concept Introduction: Lewis dot structure is also known as Lewis dot formula or electron dot structure. The bond formation between the atoms takes place due to the sharing of valence electrons of bonded atoms while the remaining electrons present in outer shell represented as lone pair of electrons.
The hybridization gives idea about the geometry of each atom. It can be checked with the below formula:
Hybridization = Number of sigma bonds + Number of lone pairs on bonded atoms.
(b)
Interpretation:The geometry of indicated atom in the given molecule with the help of hybridization needs to be determined.
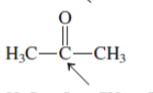
Concept Introduction: Lewis dot structure is also known as Lewis dot formula or electron dot structure. The bond formation between the atoms takes place due to the sharing of valence electrons of bonded atoms while the remaining electrons present in outer shell represented as lone pair of electrons.
The hybridization gives idea about the geometry of each atom. It can be checked with the below formula:
Hybridization = Number of sigma bonds + Number of lone pairs on bonded atoms.
(c)
Interpretation:The geometry of indicated atom in the given molecule with the help of hybridization needs to be determined.
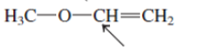
Concept Introduction: Lewis dot structure is also known as Lewis dot formula or electron dot structure. The bond formation between the atoms takes place due to the sharing of valence electrons of bonded atoms while the remaining electrons present in outer shell represented as lone pair of electrons.
The hybridization gives idea about the geometry of each atom. It can be checked with the below formula:
Hybridization = Number of sigma bonds + Number of lone pairs on bonded atoms.
(d)
Interpretation:The geometry of indicated atom in the given molecule with the help of hybridization needs to be drawn.
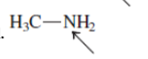
Concept Introduction: Lewis dot structure is also known as Lewis dot formula or electron dot structure. The bond formation between the atoms takes place due to the sharing of valence electrons of bonded atoms while the remaining electrons present in outer shell represented as lone pair of electrons.
The hybridization gives idea about the geometry of each atom. It can be checked with the below formula:
Hybridization = Number of sigma bonds + Number of lone pairs on bonded atoms.
(e)
Interpretation:The geometry of indicated atom in the given molecule with the help of hybridization needs to be determined.
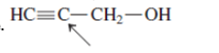
Concept Introduction: Lewis dot structure is also known as Lewis dot formula or electron dot structure. The bond formation between the atoms takes place due to the sharing of valence electrons of bonded atoms while the remaining electrons present in outer shell represented as lone pair of electrons.
The hybridization gives idea about the geometry of each atom. It can be checked with the below formula:
Hybridization = Number of sigma bonds + Number of lone pairs on bonded atoms.
(f)
Interpretation:The geometry of indicated atom in the given molecule with the help of hybridization needs to be drawn.
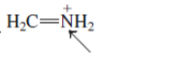
Concept Introduction: Lewis dot structure is also known as Lewis dot formula or electron dot structure. The bond formation between the atoms takes place due to the sharing of valence electrons of bonded atoms while the remaining electrons present in outer shell represented as lone pair of electrons.
The hybridization gives idea about the geometry of each atom. It can be checked with the below formula:
Hybridization = Number of sigma bonds + Number of lone pairs on bonded atoms.
Want to see the full answer?
Check out a sample textbook solution
Chapter 1 Solutions
Organic Chemistry: Structure and Function
- Explain why the two molecules below cannot inter-convert without breaking the portion of thedouble bond.arrow_forwardDraw the delocalized molecular orbitals for the following molecule. Are both bonds of the triple bond involved in the delocalized orbitals?arrow_forwardIn Chapter 6, we study a group of organic cations called carbocations. Following is the structure of one such carbocation, the tert-butyl cation. (a) How many electrons are in the valence shell of the carbon bearing the positive charge? (b) Using VSEPR, predict the bond angles about this carbon. (c) Given the bond angle you predicted in (b), what hybridization do you predict for this carbon?arrow_forward
- Use the VSEPR model to predict the bond angles around each central atom in the following Lewis structures (benzene rings are frequently pictured as hexagons, without the letter for the carbon atom at each vertex). Note that the drawings do not necessarily depict the bond angles correctly.arrow_forwardA student who missed this class needs to know how to predict the bond angles and shape of amolecule from looking at its bond-line representation. Write a concise but complete explanationfor this student.arrow_forwardDraw three contributing structures of the following compound (called guanidine) and state the hybridization of the four highlighted atoms. In which orbitals do the three lone pairs drawn reside?arrow_forward
- Consider the incomplete orbital representation of O2 , below right. a. Identify which lobes are hybrid orbitals (identify the type) and which lobes arep orbitals. b. Use dotted lines to show any bonds. c. Use up or down arrows to show electron occupation of each hybrid orbital or bond.arrow_forwardPlease indicate the orbital hybridization that exists for every carbon in the following molecules: O O ║ ║ (a) H2C=CH-CH2-C-CH3 (b) CH≡C-CH2-C-OHarrow_forwardWhat is the molecular formula?For each heteroatom (not C or H), indicate the hybridization. Also note whether each lone pair is localized (on the atom) or delocalized (through resonance).arrow_forward
- In the molecule shown label the indicated bonds with either their hybridization (sp,sp2,sp3) or the type of bond present (sigma, pi )arrow_forwardCH3+ and CH3- are two highly reactive carbon species. a. What is the predicted hybridization and geometry around each carbon atom? b. Two electrostatic potential plots are drawn for these species. Which ion corresponds to which diagram and why?arrow_forwardUse the ball-and-stick model to answer each question about carbidopa, a drug used in combination with L-dopa to treat Parkinson's disease. Determine the hybridization around each carbon atom.arrow_forward
- Chemistry: Matter and ChangeChemistryISBN:9780078746376Author:Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl WistromPublisher:Glencoe/McGraw-Hill School Pub Co
 Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning  Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning
Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning Chemistry by OpenStax (2015-05-04)ChemistryISBN:9781938168390Author:Klaus Theopold, Richard H Langley, Paul Flowers, William R. Robinson, Mark BlaserPublisher:OpenStax
Chemistry by OpenStax (2015-05-04)ChemistryISBN:9781938168390Author:Klaus Theopold, Richard H Langley, Paul Flowers, William R. Robinson, Mark BlaserPublisher:OpenStax Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning





