
Concept explainers
Interpretation:
The given ball and stick model of neral has to be converted to skeletal structure with
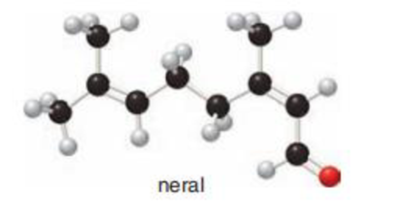
Concept Introduction:
Skeletal structures:
Skeletal structures are used for representing the organic structures in diagrammatic form. The carbon atom should have four bonds. The carbon is present at the junction of the bonds. The lone pair of electrons has to be omitted in the heteroatom. The double bond and triple bond has to be drawn. The hydrogen bonded directly to the heteroatom has to be drawn.
Functional group:
Functional group contains particular atom or group of atoms bonded in a molecule which has specific chemical and physical properties. Adding any specific functional group to compound can form a new product it is known as functionalization. The functional group contains heteroatoms and a multiple bond in a compound.
Want to see the full answer?
Check out a sample textbook solution
Chapter 10 Solutions
Principles of General, Organic, Biological Chemistry
- Draw the expanded, or complete, structural formula for the hydrocarbon represented by the line‑angle, or skeletal, structure shown.arrow_forwardWrite a condensed formula for C5H4NOClarrow_forwardAccording to Table 2.10, five constitutional isomers are possible for alkanes with six carbon atoms. Write structural and condensed formulas for these isomers.arrow_forward
- Chemistry: Matter and ChangeChemistryISBN:9780078746376Author:Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl WistromPublisher:Glencoe/McGraw-Hill School Pub Co
 Organic And Biological ChemistryChemistryISBN:9781305081079Author:STOKER, H. Stephen (howard Stephen)Publisher:Cengage Learning,
Organic And Biological ChemistryChemistryISBN:9781305081079Author:STOKER, H. Stephen (howard Stephen)Publisher:Cengage Learning, General, Organic, and Biological ChemistryChemistryISBN:9781285853918Author:H. Stephen StokerPublisher:Cengage Learning
General, Organic, and Biological ChemistryChemistryISBN:9781285853918Author:H. Stephen StokerPublisher:Cengage Learning  Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning
Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning Introductory Chemistry: An Active Learning Approa...ChemistryISBN:9781305079250Author:Mark S. Cracolice, Ed PetersPublisher:Cengage Learning
Introductory Chemistry: An Active Learning Approa...ChemistryISBN:9781305079250Author:Mark S. Cracolice, Ed PetersPublisher:Cengage Learning World of Chemistry, 3rd editionChemistryISBN:9781133109655Author:Steven S. Zumdahl, Susan L. Zumdahl, Donald J. DeCostePublisher:Brooks / Cole / Cengage Learning
World of Chemistry, 3rd editionChemistryISBN:9781133109655Author:Steven S. Zumdahl, Susan L. Zumdahl, Donald J. DeCostePublisher:Brooks / Cole / Cengage Learning





