
Concept explainers
a.
Interpretation:
The given ball and stick model to condensed or skeletal structure with
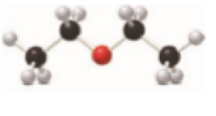
Concept Introduction:
Condensed Structures:
The compounds have a chain of atoms bonded together, other than a ring are known as condensed structure. Start writing at the left side and then ends at right side of compound. The bonds are drawn and the two electron bond lines have to be omitted.
Then lone pairs on heteroatom have to be omitted. The bonds of atoms have to be checked. The carbon atom should have four bonds.
Functional group:
Functional group contains particular atom or group of atoms bonded in a molecule which has specific chemical and physical properties. The functional group contains heteroatoms and a multiple bond in a compound.
b.
Interpretation
The given ball and stick model to condensed or skeletal structure with functional groups has to be converted.
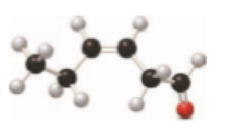
Concept Introduction:
Refer part: a.
Want to see the full answer?
Check out a sample textbook solution
Chapter 10 Solutions
Principles of General, Organic, Biological Chemistry
- Based on the structures below: i) Convert the structures above to the skeletal structure.ii) Identify the functional groups and homologous series in each compound.arrow_forwardChoose the functional group represented by the condensed structural formula (CH3)2CHCHO.arrow_forwardDraw 8 isomers (with condensed formula and skeletal structure) with the formula C8H18.arrow_forward
- Please convert the following compound from its condensed structural formula into its expanded formula (draw the compound). (CH3)2CHCOOCH3arrow_forwardConvert each condensed formula to a complete structure with lone pairs on heteroatoms. a. CH 3(CH 2) 8CH 3 c. CH 3CCl 3 e. (CH 3) 2CHCH 2NH 2 b. CH 3(CH 2) 4OH d. CH 3(CH 2) 4CH(CH 3) 2arrow_forwarddraw expanded,condensed and skeletal structure of atenololarrow_forward
 Introductory Chemistry: An Active Learning Approa...ChemistryISBN:9781305079250Author:Mark S. Cracolice, Ed PetersPublisher:Cengage Learning
Introductory Chemistry: An Active Learning Approa...ChemistryISBN:9781305079250Author:Mark S. Cracolice, Ed PetersPublisher:Cengage Learning Principles of Modern ChemistryChemistryISBN:9781305079113Author:David W. Oxtoby, H. Pat Gillis, Laurie J. ButlerPublisher:Cengage Learning
Principles of Modern ChemistryChemistryISBN:9781305079113Author:David W. Oxtoby, H. Pat Gillis, Laurie J. ButlerPublisher:Cengage Learning Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning
Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning- Chemistry: Matter and ChangeChemistryISBN:9780078746376Author:Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl WistromPublisher:Glencoe/McGraw-Hill School Pub Co



