
a.
Interpretation:
The compounds butane or isobutane has to be represented.
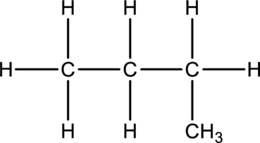
Concept Introduction:
Butane and Isobutane:
In butane structure there will be four carbon atoms present in the straight chain. The carbon present in one continuous chain.
In isobutane there will be three carbon atoms present in a row and one carbon bond is bonded to middle carbon. This
b.
Interpretation
The compounds whether butane or isobutane has to be identified.
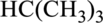
Concept Introduction:
Refer part: a.
c.
Interpretation:
The compounds either butane or isobutane has to be represented.
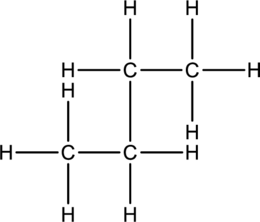
Concept Introduction:
Refer part: a.
d.
Interpretation:
The compounds butane or isobutane has to be represented.
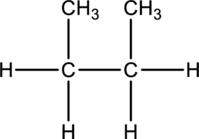
Concept-Introduction:
Refer part: a.
Want to see the full answer?
Check out a sample textbook solution
Chapter 10 Solutions
Principles of General, Organic, Biological Chemistry
- Identify each compound according to its functional group:arrow_forwarda. What is the chemical structure of 2,6-dichloroindophenol, circle functional groups differentthan alkane, alkene, alkyne? b. Is it polar or nonpolar? _______________________ c. What is its water solubility in g/L? ___________arrow_forwardBy adding tetraethyl lead to iso-octane: A. Exhaust smoke can be reduced B. Knocking can be completely suppressed C. Octane number above 100 can be obtained D. Specific fuel consumption can be reducedarrow_forward
- put the correct substituentarrow_forward1. The following are isomers of each other I. pentane II. 2,2,-dimethylpropane III. 2-methylbutaneIV. 2,3-dimethylhexane A. Alkanes are nonpolar. B. Alkanes are non-flammable. C. Every carbon in an alkane has four bonds. D. Alkanes contain only C and H atoms. E. Alkanes do not contain carbon-carbon double or triple bonds. 1b. Which of these is a weak base? A. NaOH B. H3PO4 C. CH3COOH D. NaHCO3 E. NH4NO3 1c. Which of these pairs are formed when NaHCO3 (baking soda) is dissolved in water? NaHCO3 → ______ + ______. A. NaOH + CO2 B. Na+ +HCO3- C. NaH+ +CO3- D. Na + HCO3 E. NaH- + CO3+ 1D. Which of these statements about water is(are) incorrect? A. It has a pH of 7.0 at ambient temperatures. B. It contains no ions. C. It is neutral. D. It can act as a proton donor. E. It can act as a proton acceptor.arrow_forward(a)({)-hexane-3,4-diol, draw any enantiomer.arrow_forward
 Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning

 World of Chemistry, 3rd editionChemistryISBN:9781133109655Author:Steven S. Zumdahl, Susan L. Zumdahl, Donald J. DeCostePublisher:Brooks / Cole / Cengage Learning
World of Chemistry, 3rd editionChemistryISBN:9781133109655Author:Steven S. Zumdahl, Susan L. Zumdahl, Donald J. DeCostePublisher:Brooks / Cole / Cengage Learning Chemistry: An Atoms First ApproachChemistryISBN:9781305079243Author:Steven S. Zumdahl, Susan A. ZumdahlPublisher:Cengage Learning
Chemistry: An Atoms First ApproachChemistryISBN:9781305079243Author:Steven S. Zumdahl, Susan A. ZumdahlPublisher:Cengage Learning





