
Concept explainers
Interpretation:
The reason has to be explained for the given compound does not undergo a nucleophilic substitution reaction by either SN1 or SN2 mechanisms.
Concept introduction:
SN1 reaction:
The alcohol is reaction with acids like hydrochloric acid or hydrobromic acid which yield the corresponding carbocation intermediate, this carbocation intermediate undergoes substitution reaction which yields the corresponding substitution product.
Tertiary alcohols undergo substitution very fast than the secondary alcohols because tertiary carbocation is more stable than the secondary carbocation than the primary carbocation. Primary alcohol is less stable therefore it won’t undergo SN1 substitution reaction.
SN2 reaction:
Tosylated compound is reaction with sodium methoxide which undergoes again SN2 type of reaction, the methoxide ion attacks the carbon atom through the back side and provides Inverse configuration of methoxy compound. This is shown below,
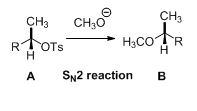
Given information:
The given compound is shown below,
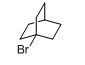
Trending nowThis is a popular solution!

Chapter 11 Solutions
ORGANIC CHEM.(LL)-W/OWL V2 >CUSTOM<
- List the following order of increasing reactivity in an SN1 reactionarrow_forwardWhat are the relative rates of the following nucleophilic substitution reaction below.arrow_forwardThe compound below undergoes electrophilic aromatic substitution to form a monosubstituted product. At which position will the electrophile going to attack?arrow_forward
- Trimethyl Phosphite reacts with bromomethane to afford the product shown below via a two-step mechanism. Draw a detailed mechanism for this reaction using arrows to show electron flow (both mechanistic steps are SN2 reactions).arrow_forwardIdentify which substitution mechanism best fits the following statement: The reaction proceeds through a concerted mechanism. A) SN1 B) SN2arrow_forwardWhich compound shown below would undergo nucleophilic substitution via addition/elimination the most quickly?arrow_forward
- Both menthyl chloride and neomenthyl chloride (structures shown below) undergo E2 elimination in the presence of EtONa in ethanol. Provide the possible product(s). Which substrate reacts faster in term of overall reaction rate? Explain why.arrow_forward3-Bromocyclohexene is a secondary halide, and benzyl bromide is a primary halide. Both halides undergo SN1 substitution about as fast as most tertiary halides. Use resonance structures to explain this enhanced reactivity.arrow_forwardWhich of the following reacts fastest with methanol by the SN1 mechanism?arrow_forward
- A student in Ochem proposed the following synthesis. Explain the problem with this approach and provide the necessary reagents to convert halohydrin A to alkene B (more than one step is required) in addition provide the product after each step. Provide the mechanism and expected actual final product from the reaction of halohydrin A with LDAarrow_forwardComplete the following reactions by supplying the structure of the product/s. Identify the mechanism involved (E1, E2, SN1, SN2). A. Zaitsev product B. Anti - Zaitsev product C. Mechanismarrow_forwardThese two compounds shown go through SN1 substitution at a speed similar to some tertiary halides, even though the following compounds represent primary and secondary halides. Explain the enhanced reactivity via resonance structures and pushing arrows.arrow_forward

 Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning


