
Interpretation:
The eight diastereomers of 1,2,3,4,5,6-hexachlorocyclohexane has to be drawn and the reason has to be explained for One isomer loses HCl in an E2 reaction nearly 1000 times more slowly than the others.
Concept introduction:
Conformations: Rotation about C-C single bonds allows a compound to adopt a variety of possible three-dimensional shapes.
Drawing Axial and Equatorial substituents:
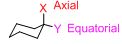
Each carbon in cyclohexane can bear two substituents. One group is said to occupy an axial position, which is parallel to a vertical axis passing through the center of the ring. the other group is said to occupy an equatorial position, which is positioned approximately along the equator of the ring.
E2 elimination:
Want to see the full answer?
Check out a sample textbook solution
Chapter 11 Solutions
ORGANIC CHEM.(LL)-W/OWL V2 >CUSTOM<
- Ketones react with alcohols to yield products called acetals. Why does the all-cis isomer of 4-tert-butyl-1,3-cyclohexanediol react readily with acetone and an acid catalyst to form an acetal, but other stereoisomers do not react? In formulating your answer, draw the more stable chair conformations of all four stereoisomers and the product acetal for each one.arrow_forwardPlease predict the products for each of the following reactions. *HBr in red* Clearly show the regiochemistry (Markovnikov vs anti-Markovnikov) and stereochemistry (syn- vs anti- or both). If a mixture of enantiomers is formed, please draw all the enantiomers. Choose an answer: A, B, C, or Darrow_forwardGiven that the free energy of the twist-boat conformer of cyclohexane is 5.3 kcal/mol greater than that of the chair conformer, calculate the percentage of twist-boat conformers present in a sample of cyclohexane at 25 °C.arrow_forward
- We saw that the energy cost of an axial methyl is 1.8 kcal/mol; therefore, we might expect cis-1,3-dimethylcyclohexane to have chair conformations with a difference in totalenergy of 3.6 kcal/mol. 1. Is the calculated energy difference in cis-1,3-dimethylcyclohexane higher or lower than expected? Propose an explanation for the difference between the expected energy difference and the calculated energy difference in cis-1,3-dimethylcyclohexane. (Limit your answer to 10 words or fewer) 2. Recall that an axial methyl is typically worth around 1.8 kcal/mol. Propose an explanation for the difference in the value above for 1-methyltetrahydropyran. (Limit your answer to 10 words or less) 3. Would you expect the difference in strain energy between the chairs of 5-methyl-1,3-dioxane to be greater than or less than that of 1-methyltetrahydropyran? Is it GREATER THAN or LESS THAN?arrow_forwardGiven that the change in free energy between axial and equatorial chlorine is 0.52 kcal.mol-1 , what is the difference in energy between the two chair conformations of 1,2,3,5-tetrachlorocyclohexane?What is the percent of the most stable chair conformation in solution at 298K?arrow_forwardThe alkene shown undergoes bromination. (a) Draw the product(s) of bromination of this compound, including all expected stereoisomers (if any). Use wedge‑and‑dash bonds to designate the stereochemistry at any chirality centers, and make sure to draw an explicit hydrogen if a chirality center has one. (b) Characterize the starting alkene as having the E or Z configuration. (c) characterize the product(s).arrow_forward
- What stereoisomers are obtained from hydroboration–oxidation of the following compounds? Assign an R or S configuration to each asymmetric center. a. cyclohexene b. 1-ethylcyclohexene c. cis-2-butene d. (Z)-3,4-dimethyl-3-hexenearrow_forwardWhen the given alkene undergoes hydroboration followed by oxidation, which would be produced in regards to the products stereochemistry a. A mix of diastereomers b. A single enantiomer c. A racemic mixture d. An achiral productarrow_forwardWhat is the degree of unsaturation present in D.D.Tarrow_forward
- What organic product is formed when 1‑methylcyclopentene is treated with NMMO in the presence of H2O and a catalytic amound of OsO4? Clearly show stereochemistry by drawing a wedge and dashed bond for each chiral carbon. Only draw one stereoisomer if more than one can be formed.arrow_forward1. Estimate the relative stabilities of the 4 conformationseclipsed and eclipsed n-pentane by drawing the corresponding Newman projections in descending order according to their stability. Place the most stable or lowest energy on the left. 2. Explain what the difference in energy between the most stable and the least stable is due to. 3. What is the difference in stability between the two staggered conformations?arrow_forwardFollowing is a planar hexagon representation of L-fucose, a sugar component of the determinants of the A, B, O blood group typing. For more on this system of blood typing, see Chemical Connections: A, B, AB, and O Blood Group Substances in Chapter 25. (a) Draw the alternative chair conformations of L-fucose. (b) Which of them is more stable? Explain.arrow_forward

 Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning


