
Interpretation:
The Newman projection has to be drawn and the reason has to be explained for the formation of more trans product.
Concept introduction:
Conformations: Rotation about C-C single bonds allows a compound to adopt a variety of possible three-dimensional shapes.
Newman projections: The new conformations of compounds can be drawn and analyzed by Newman projections. A Newman projection visualizes different conformations of Carbon-carbon
The angle between two hydrogens of a Newman projection is called as dihedral angle or torsional angle. This dihedral angle changes as the C-C bond rotates. Two conformations with special attentions are staggered and eclipsed conformation. Staggered conformation is the lowest in energy and the eclipsed conformation is the highest in energy.
For example,

Anti-conformation: The conformation with a dihedral angle of is called anti-conformation.
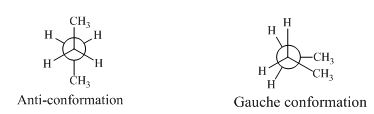
The two methyl groups achieve maximum separation from each other. In other, methyl groups are closer to each other; their electron clouds are repelling each other, causing an increase in energy. This unfavorable interaction is called gauche interaction.
E2 elimination:
Trending nowThis is a popular solution!

Chapter 11 Solutions
ORGANIC CHEM.(LL)-W/OWL V2 >CUSTOM<
- Which of the two isomers would you expect to undergo E2 elimination faster? trans-1-bromo- 4-tert-butylcyclohexane or cis-1-bromo-4-tert-butylcyclohexane? Draw each molecule in its more stable chair conformation. Provide an explanation to your answer.arrow_forwardDraw the structure consistent with following description. (3E,5Z)-nona-3,5-diene in the s-cis conformationarrow_forwardFor each reaction, decide whether substitution or elimination (or both) is possible, andpredict the products you expect. Label the major products.(a) 1@bromo@1@methylcyclohexane + NaOH in acetonearrow_forward
- Provide the complete mechanism using Curved Arrow Formalism for the reaction of 1-chloro-1-methylcyclopentane treated with NaOMe in MeOH. If more than one product is formed, which is the major, minor, and very minor product? Is the reaction SN1, SN2, E1, or E2. I appreciate the help on this question.arrow_forwardCis-1-bromo-4-isopropylcyclohexane undergoes E2 elimination, with sodium hydroxide, 5000 times faster than does trans-1-bromo-4-isopropylcyclohexane. Fully explain this reactivity difference using mechanisms which clearly show the transition state. You should draw both of these isomers in the chair form.arrow_forwardConsider the following statement in reference to SN1, SN2, E1, and E2 reactions of haloalkanes. To which mechanism(s), if any, does the statement apply? Involves a carbocation intermediatearrow_forward
- Five-membered aromatic heterocycles with one heteroatom undergo electrophilic substitutions preferentially at the α-position (C-2 and C-5) rather than at the β-position (C-3 and C-4). Explain?arrow_forwardWe'll see in the next chapter that the stability of carbocations depends on the number of alkyl substituents attached to the positively charged carbon - the more alkyl substituents there are, the more stable the cation. Which of the two carbocations in each pair is more stable? If they are of equal stablility, specify this.arrow_forwardAlthough there are nine stereoisomers of 1,2,3,4,5,6-hexachlorocyclohexane, one stereoisomer reacts 7000 times more slowly than any of the others in an E2 elimination. Draw the structure of this isomer and explain why this is so.arrow_forward
 Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning

