
Interpretation:
The formation of
Concept introduction:
Elimination reaction: An elimination reaction is removal of two substituents in a molecule and forms alkene. An elimination reaction is one or two-step process which based on the mechanism when two substituents removed from the molecule in single step is called E2 reaction. When two substituents are removed from the molecule in two steps is called E1 reaction.
E2 elimination:Alkyl halide forms an alkene from the abstraction of the proton from the β-carbon atom followed by elimination of the bromine in a single step.
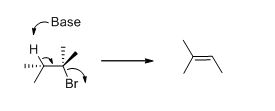
Given information:
The given compound is shown below,
Trending nowThis is a popular solution!

Chapter 11 Solutions
ORGANIC CHEM.(LL)-W/OWL V2 >CUSTOM<
- Show the stereochemistry of the epoxide (see Problem 11-31) you would obtain by formation of a bromohydrin from trans-2-butene, followed by treatment with base.arrow_forwardShow the carbocation pathway for the given reactions below. a. (2E)-4-methyl-2-pentene + HCl b. cyclopentene + H2O (acid catalyzed) c. 1-butene + CH3OHarrow_forwardWhat alkyl halide would produce methylenecyclohexane after an E2 reacyion?arrow_forward
- Which of the statements below are correct about characteristics of an E2 reaction? i) Forms alkene product ii) Requires strong base iii) Minimum of one step iv) Concerted mechanismarrow_forwardFor each reaction, show the generation of the electrophile and predict the products.(a) benzene + cyclohexene + HF (b) tert@butyl alcohol + benzene + BF3(c) tert@butylbenzene + 2@methylpropene + HFarrow_forwardWhat will be the major product of the reaction of 2-methyl-2-butene with each of the following reagents? a) Br2/H2O b)BH3/THF followed by H2O/Hydeoxyl ionarrow_forward
- What is the major product obtained when each of the following compounds undergoes an E2 reaction with methoxide ion? Show the configuration of the product. Does the product obtained depend on whether you start with the R or S enantiomer of the reactant?arrow_forwardHow does each of the following changes affect the rate of an E1 reaction? a. doubling [RX] b. doubling [B:] c. changing the halide from (CH3)3CBr to CH3CH2CH2Br d. changing the leaving group from Cl− to Br− e. changing the solvent from DMSO to CH3OHarrow_forwardHow does each of the following changes affect the rate of an E1 reaction? a.doubling [RX] b.doubling [B:] c.changing the halide from (CH3)3CBr to CH3CH2CH2Br d.changing the leaving group from Cl− to Br− e. changing the solvent from DMSO to CH3OHarrow_forward
- Which of the reagents listed below would efficiently accomplish the transformation of CH 3CH 2CH=CHCH 2CH 2CHO into CH 3CH 2CH 2CH 2CH 2CH 2CH 2OH? A. KMnO4 B. NaBH4 C. Br2 in CCl4 D. H2, Ni E. Two of the abovearrow_forwardfrom the list of reagents select those that carry out the transformation above the list.please make sure the corrosponding letters are in the correct order for the reaction starting on the left.i.e. cd = 1. LiAlH4 then H3O+, 2. H2, Pd/Carrow_forward1. What products are formed from a reaction flask that contains 2-bromopentane in ethanolic solution of sodium ethoxide? 2. What reaction pathway does the above reaction follow? 3. What is responsible for the major and minor products formed from the above reaction?arrow_forward

 Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning


