
Essential Organic Chemistry (3rd Edition)
3rd Edition
ISBN: 9780321937711
Author: Paula Yurkanis Bruice
Publisher: PEARSON
expand_more
expand_more
format_list_bulleted
Concept explainers
Textbook Question
Chapter 12, Problem 31P
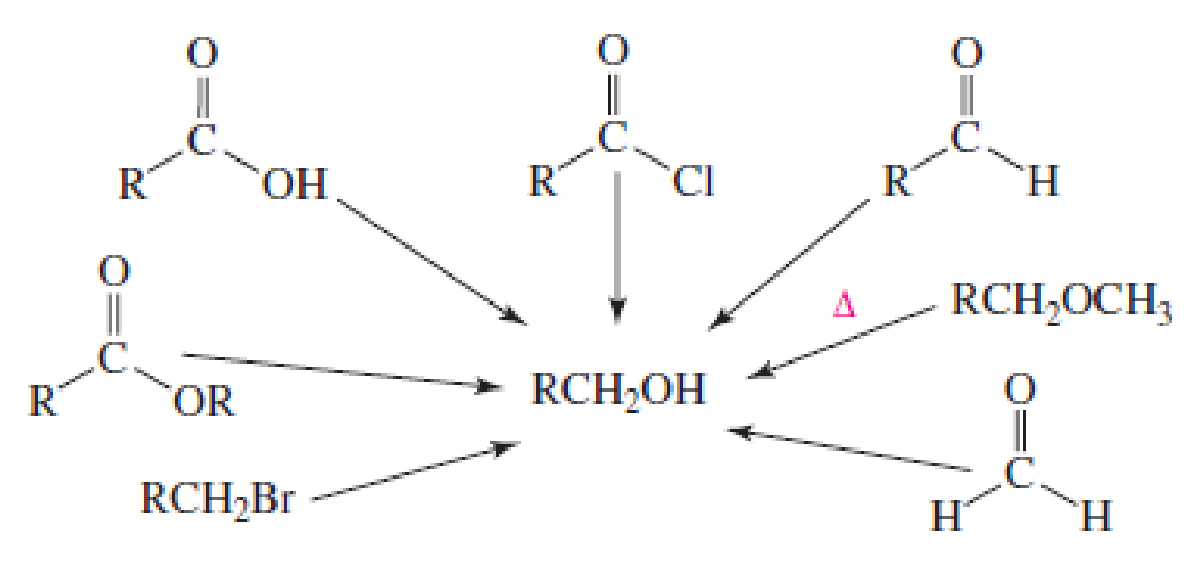
Show the reagents required to form the primary alcohol in each of the following reactions:
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
Of the following reagents, which could be used to make the alcohol shown?
1. H2O/H30+
2. 1) Hg(OAc)2 H2O:2) NaBH4
3. ) 1) BH3;2)H2O2, OH-
Which of the following forms a 1° alcohol when reacted with Grignard reagent?
A. ketone
B. carbon dioxide
C. formaldehyde
D. acyl halide
Predict the products of the sulfuric acid-catalyzed dehydration of the following alcohols. When more than one product is expected, label the major and minor products.
(a) 2-methylbutan-2-ol (b) pentan-1-ol
Chapter 12 Solutions
Essential Organic Chemistry (3rd Edition)
Ch. 12.1 - Prob. 1PCh. 12.1 - Name the following compounds:Ch. 12.1 - Prob. 3PCh. 12.1 - Prob. 4PCh. 12.2 - Prob. 5PCh. 12.4 - Draw the structure for each of the following: a....Ch. 12.4 - Prob. 7PCh. 12.5 - What products are formed when the following...Ch. 12.5 - Prob. 9PCh. 12.5 - Prob. 10P
Ch. 12.5 - Prob. 12PCh. 12.5 - Write the mechanism for the reaction of acetyl...Ch. 12.5 - Prob. 14PCh. 12.6 - Prob. 16PCh. 12.7 - Prob. 17PCh. 12.7 - Prob. 18PCh. 12.7 - Prob. 19PCh. 12.8 - Prob. 20PCh. 12.8 - Prob. 21PCh. 12.8 - Prob. 22PCh. 12.9 - Which of the following are a. hemiacetals? b....Ch. 12.9 - Prob. 24PCh. 12.9 - Prob. 25PCh. 12.10 - Prob. 26PCh. 12.11 - Prob. 27PCh. 12 - Draw the structure for each of the following: a....Ch. 12 - Prob. 29PCh. 12 - List the following compounds in order from most...Ch. 12 - Show the reagents required to form the primary...Ch. 12 - Fill in the boxes:Ch. 12 - Indicate how the following compounds could be...Ch. 12 - Prob. 34PCh. 12 - Prob. 35PCh. 12 - Prob. 36PCh. 12 - Prob. 37PCh. 12 - Prob. 38PCh. 12 - Prob. 39PCh. 12 - Show two ways the following compound could be...Ch. 12 - Prob. 41PCh. 12 - Prob. 42PCh. 12 - Prob. 43PCh. 12 - Prob. 44PCh. 12 - Prob. 45PCh. 12 - Prob. 46PCh. 12 - Prob. 47PCh. 12 - Prob. 48PCh. 12 - Prob. 49PCh. 12 - Prob. 50PCh. 12 - Prob. 51PCh. 12 - Indicate how the following compounds could be...Ch. 12 - Prob. 53P
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- Starting with acetylene and ethylene oxide as the only sources of carbon atoms, show how to prepare these compounds. (a) 3-Butyn-1-ol (b) 3-Hexyn-1,6-diol (c) 1,6-Hexanediol (d) (Z)-3-Hexen-1,6-diol (e) (E)-3-Hexen-1,6-diol (f) Hexanedialarrow_forwardGive the structure of the principal product(s) when each of the following alcohols reacts with (1) Na2Cr2O7/H2SO4, (2) PCC, (3) DMP, and (4) 1 equiv NaOCl-TEMPO. (a) 4-hydroxydecanal (b) 1-methylcyclohexan-1,4-diolarrow_forwardPredict the products of the sulfuric acid-catalyzed dehydration of the following alcohols.When more than one product is expected, label the major and minor products. pentan-1-olarrow_forward
- Predict the products of the sulfuric acid-catalyzed dehydration of the following alcohols. When more than one product is expected, label the major and minor products. (a) pentan-2-ol (b) 1-isopropylcyclohexanol (c) 2-methylcyclohexanolarrow_forwardShow how you would use a simple chemical test to distinguish between the followingpairs of compounds. Tell what you would observe with each compound.1. hexan-1-ol and cyclohexanol2. allyl alcohol and propan-1-olarrow_forwardWhat products would you expect from the oxidation of the following alcohols? 1. alcohol (a) 2. alcohol (b) 3. alcohol (c)arrow_forward
- 1. (a) Which compound would have the highest boiling point? benzophenone toluene acetophenone benzaldehyde (b) Which of the reagents listed below can convert a primary alcohol to an aldehyde? H2CrO4 DMSO, (COCl)2, Et3N AgNO3 KMnO4-, NaOH (c) Oxidation of alcohols involving the use of DMSO and (COCl)2 is also known as which of the following? Jones Tollens Baeyer-Villiger Swernarrow_forward1. O-hydroxybenzoic acid is a major product formed with phenol and which other reactant/s I-primary alcohol II-sodium hydroxide III-water IV-carbon dioxide A.I and III B. I and IV C. II and III D. II and IVarrow_forwardShow reagents and conditions by which you could prepare each of the following compounds from cyclohexanone.arrow_forward
- Which of the following starting materials would yield the following product? a. 2-hexanol b. 2-heptanol c. 1-heptanol d. 1-hexanolarrow_forwardPropose a structural formula for the products when each of the following our Keynes is treated with H2O/H2S04. Why are two products formed and part B but only one in parts a and c. a. 1 Hexene gives one alcohol with a molecular formula of C6 H 14 O. b. Two hexene gifts to our calls each with a molecular formula of C6 H 14 O. c. Three hexene gives one alcohol with a molecular formula of C6 H 12 O.arrow_forwardWhat are the conditions needed to promote an SN2 type of reaction for the synthesis of an alcohol? for example would you use a primary, secondary, or teritiary alkyl halide as a starting material for the synthesis of an alcohol?arrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you

 Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning
Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning


Organic Chemistry
Chemistry
ISBN:9781305580350
Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. Foote
Publisher:Cengage Learning

Introduction to General, Organic and Biochemistry
Chemistry
ISBN:9781285869759
Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar Torres
Publisher:Cengage Learning
How to Design a Total Synthesis; Author: Chemistry Unleashed;https://www.youtube.com/watch?v=9jRfAJJO7mM;License: Standard YouTube License, CC-BY