
Concept explainers
(a)
Interpretation:
The number of stereoisomers has to be identified when the reaction of 2-pentanone with ethyl magnesium bromide followed by the addition of dilute acid.
Concept introduction:
The Grignard reaction:
Alkyl, vinyl, or aryl-magnesium halides (
Grignard reagent is reaction with carbonyl compound such as
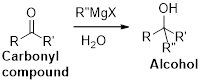
Stereoisomers: Stereoisomers are molecules that have the same molecular formula and they differ only in arrangement of atom in three-dimensional space.
(b)
Interpretation:
The number of stereoisomers has to be identified when the reaction of 2-pentanone with methyl magnesium bromide followed by the addition of dilute acid.
Concept introduction:
The Grignard reaction:
Alkyl, vinyl, or aryl-magnesium halides (
Grignard reagent is reaction with carbonyl compound such as aldehyde or ketone, produces corresponding alcohol is the product and it is the one of the carbon – carbon bond forming reaction.
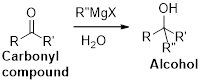
Stereoisomers: Stereoisomers are molecules that have the same molecular formula and they differ only in arrangement of atom in three-dimensional space.
Want to see the full answer?
Check out a sample textbook solution
Chapter 12 Solutions
Essential Organic Chemistry (3rd Edition)
- What product is obtained from the reaction of excess benzene with isobutyl chloride + AlCl3 ?arrow_forwardWhat is the major product(s) of each of the following reactions? a. bromination of p-methylbenzoic acid b. chlorination of o-benzenedicarboxylic acid c. bromination of p-chlorobenzoic acid d. nitration of p-fluoroanisole e. nitration of p-methoxybenzaldehyde f. nitration of p-tert-butylmethylbenzenearrow_forwardFrom which starting materials can the product formed as a result of the following reaction be synthesized? A. Acetophenone and butanal B. Benzaldehyde and 3-pentanone C. Benzaldehyde and 2-pentanone D. Acetophenone and 2-butanonearrow_forward
- Explain why the ortho-substituted 2-nitrophenol in 30-40% yield and the para-substituted 4-nitrophenol in 15% yield have different boiling points and in doing so, explain which isomer has the higher boiling point and which one has the lower boiling point?arrow_forwardThe reaction of a nitrile with an alcohol in the presence of a strong acid forms an N-substituted amide. This reaction, known as the Ritter reaction, doesnot work with primary alcohols. a. Propose a mechanism for the Ritter reaction.b. Why does the Ritter reaction not work with primary alcohols?c. How does the Ritter reaction differ from the acid-catalyzed hydrolysis of a nitrile to form an amide?arrow_forwardWhat product is obtained from the reaction of excess benzene with dichloromethane + AlCl3 ?arrow_forward
- How many stereoisomers are obtained from the reaction of 3-methyl-2-butanone with each of the following organometallic reagents followed by the addition of dilute acid? Name each stereoisomer. ethyllithium methylmagnesium bromidearrow_forward3′-chloro-4′-methoxyacetophenone is less or more polar than 4-methoxyacetophenone? explain why (WITHOUT DRAWINGS)arrow_forwardThe reaction of a nitrile with an alcohol in the presence of a strong acid forms an N-substituted amide. This reaction, known as the Ritter reaction, doesnot work with primary alcohols. a. Why does the Ritter reaction not work with primary alcohols? b. Provide an explanation for why an amide is less susceptible to nucleophilic attack than its corresponding ester.arrow_forward
- Reduction of Benzophenone by Sodium borohydride.arrow_forwardGive reasons :(a) n-Butyl bromide has higher boiling point than f-butyl bromide.(b) Racemic mixture is optically inactive.(c) The presence of nitro group (-NO2) at o/p positions increases the reactivity of haloarenes towards nucleophilic substitution reactions.arrow_forward3′-chloro-4′-methoxyacetophenone has a higher melting point than 4-methoxyacetophenone. explain why?arrow_forward

 Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning


