
Essential Organic Chemistry (3rd Edition)
3rd Edition
ISBN: 9780321937711
Author: Paula Yurkanis Bruice
Publisher: PEARSON
expand_more
expand_more
format_list_bulleted
Question
Chapter 12.8, Problem 22P
(a)
Interpretation Introduction
Interpretation:
The carbonyl compound should be identified for the given amino acid preparation.
Concept introduction:
Amino acid:
An amino acid is an organic molecule and it consists of
The general formula for the amino acid is given below,
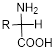
(b)
Interpretation Introduction
Interpretation:
The carbonyl compound should be identified for the given amino acid preparation.
Concept introduction:
Amino acid:
An amino acid is an organic molecule and it consists of amine group
The general formula for the amino acid is given below,
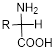
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
Predict the products obtained from the reaction of triolein with the following reagents.(a) NaOH in water (b) H2 and a nickel catalyst (c) Br2 in CCl4
Which of the following substituent is an ortho/para-directing activator and an ortho/para-directing deactivator?
a
amino group
b
halogen group
c
nitro group
d
carboxyl group
1. Give the order of basicity of alkaloids based on the R-groups attached to the amino functional group. Based on your knowledge in organic chemistry, explain the reason why one is more or less basic as compared to the other.
Chapter 12 Solutions
Essential Organic Chemistry (3rd Edition)
Ch. 12.1 - Prob. 1PCh. 12.1 - Name the following compounds:Ch. 12.1 - Prob. 3PCh. 12.1 - Prob. 4PCh. 12.2 - Prob. 5PCh. 12.4 - Draw the structure for each of the following: a....Ch. 12.4 - Prob. 7PCh. 12.5 - What products are formed when the following...Ch. 12.5 - Prob. 9PCh. 12.5 - Prob. 10P
Ch. 12.5 - Prob. 12PCh. 12.5 - Write the mechanism for the reaction of acetyl...Ch. 12.5 - Prob. 14PCh. 12.6 - Prob. 16PCh. 12.7 - Prob. 17PCh. 12.7 - Prob. 18PCh. 12.7 - Prob. 19PCh. 12.8 - Prob. 20PCh. 12.8 - Prob. 21PCh. 12.8 - Prob. 22PCh. 12.9 - Which of the following are a. hemiacetals? b....Ch. 12.9 - Prob. 24PCh. 12.9 - Prob. 25PCh. 12.10 - Prob. 26PCh. 12.11 - Prob. 27PCh. 12 - Draw the structure for each of the following: a....Ch. 12 - Prob. 29PCh. 12 - List the following compounds in order from most...Ch. 12 - Show the reagents required to form the primary...Ch. 12 - Fill in the boxes:Ch. 12 - Indicate how the following compounds could be...Ch. 12 - Prob. 34PCh. 12 - Prob. 35PCh. 12 - Prob. 36PCh. 12 - Prob. 37PCh. 12 - Prob. 38PCh. 12 - Prob. 39PCh. 12 - Show two ways the following compound could be...Ch. 12 - Prob. 41PCh. 12 - Prob. 42PCh. 12 - Prob. 43PCh. 12 - Prob. 44PCh. 12 - Prob. 45PCh. 12 - Prob. 46PCh. 12 - Prob. 47PCh. 12 - Prob. 48PCh. 12 - Prob. 49PCh. 12 - Prob. 50PCh. 12 - Prob. 51PCh. 12 - Indicate how the following compounds could be...Ch. 12 - Prob. 53P
Knowledge Booster
Similar questions
- Features of the structure of imino acids. Formation of amides from amino acids.arrow_forwardα-Amino acids can be prepared by treating an aldehyde with ammonia/trace acid, followed by hydrogen cyanide, followed by acid-catalyzed hydrolysis. Draw the structures of the two intermediates formed in this reaction.arrow_forwardd-Glucuronic acid is found widely in plants and animals. One of its functions is to detoxify poisonous HO-containing compounds by reacting with them in the liver to form glucuronides. Glucuronides are water soluble and, therefore, readily excreted. After ingestion of a poison such as turpentine or phenol, the glucuronides of these compounds are found in urine. Draw the structure of the glucuronide formed by the reaction of beta-d-glucuronic acid and phenol.arrow_forward
- Explain the observed difference in the pKa values of the conjugate acids of amines A and B.arrow_forwardThe hydrolysis of the ester shown here is catalyzed by morpholine. Explain how morpholine catalyzes the reaction. (Hint: The pKa of the conjugateacid of morpholine is 9.3, so morpholine is too weak a base to function as a base catalyst.)arrow_forwardthe scheme shown consists of key steps in the acidic hydrolysis of an amide. Complete the mechanism by drawing curved arrowsin each steparrow_forward
- d-Glucuronic acid is found widely in plants and animals. One of its functions is to detoxify poisonous HO-containing compounds by reacting with them inthe liver to form glucuronides. Glucuronides are water soluble and, therefore, readily excreted. After ingestion of a poison such as turpentine or phenol, theglucuronides of these compounds are found in urine. Draw the structure of the glucuronide formed by the reaction of β-d-glucuronic acid and phenol.arrow_forwardWhich of these 3 compounds is the least basic: aniline, 2-propylamine and 1-(2-aminophenyl)ethenone.arrow_forwardHow do you synthesize the amino acid Phenylalanine from benzaldehyde as the starting compound? >please write out all the steps and reagents involvedarrow_forward
- The structure shown below is an intermediate in the synthesis of which biogenic amine? Use arrows to show how the next intermediate in this reaction is formed, and draw the structure of that intermediate.arrow_forwardarrange the following compound types in order of decreasing ease of hydrolysis: acid halides, acid anhydrides, esters, and amides. Use > in your arrangement.arrow_forwardExplain below ? What happens when bromine of the alpha carbon of a carboxylic acid is treated with a weak or strong base?arrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning

Organic Chemistry: A Guided Inquiry
Chemistry
ISBN:9780618974122
Author:Andrei Straumanis
Publisher:Cengage Learning

Organic Chemistry
Chemistry
ISBN:9781305580350
Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. Foote
Publisher:Cengage Learning