
Concept explainers
(a)
Interpretation:
The three different sets of reagents (each set containing of a carbonyl compound and Grignard reagent) have to be used to prepare each of the following tertiary alcohols.
Concept introduction:
The Grignard reaction:
Alkyl, vinyl, or aryl-magnesium halides (
Grignard reagent is reaction with carbonyl compound such as
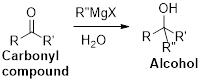
(b)
Interpretation:
The three different sets of reagents (each set containing of a carbonyl compound and Grignard reagent) have to be used to prepare each of the following tertiary alcohols.
Concept introduction:
The Grignard reaction:
Alkyl, vinyl, or aryl-magnesium halides (
Grignard reagent is reaction with carbonyl compound such as aldehyde or ketone, produces corresponding alcohol is the product and it is the one of the carbon – carbon bond forming reaction.
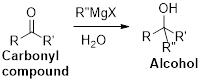
Want to see the full answer?
Check out a sample textbook solution
Chapter 12 Solutions
Essential Organic Chemistry (3rd Edition)
- Which of the following will not form an alcohol when reacted with 1 equivalent of Grignard reagent? A. acyl halide B. aldehyde C. ketone D. epoxidearrow_forwardShow reagents and conditions by which you could prepare each of the following compounds from cyclohexanone.arrow_forwardWhich of the following statements is true10. As the alkyl chain increases in molecular weight among primary alcohol, the solubility in water increases. 11. An ethoxide ion exhibits greater proton-accepting property compared to an ethyloxonium ion. 12. Phenol is more acidic than cyclohexyl alcohol due to the electron-donating ability of phenolic oxygen.arrow_forward
- Convert benzene into each compound. You may also use any inorganic reagents and organic alcohols having three carbons or fewer. One step of the synthesis must use a Grignard reagent.arrow_forwardConvert benzene into each compound. You may also use any inorganic reagents and organic alcohols having four or fewer carbons. One step of the synthesis must use a Grignard reagent.arrow_forwardWhich of the following alcohols will have NO REACTION upon oxidation with potassium dichromate in an acidic solution (Jones Oxidation)? 2-methyl-2-pentanol 3-methyl-1-butanol All of the given 4-methyl-2-pentanolarrow_forward
- Give the structure of the principal product(s) when each of the following alcohols reacts with (1) Na2Cr2O7/H2SO4, (2) PCC, (3) DMP, and (4) 1 equiv NaOCl-TEMPO. (a) 4-hydroxydecanal (b) 1-methylcyclohexan-1,4-diolarrow_forwardWhat is the major product obtained when each of the following alcohols is heated in the presence of H2SO4?arrow_forwardWhich of the following alcohols will yield a carboxylic acid product upon oxidation with potassium dichromate in an acidic solution (Jones Oxidation)? 2-methyl-2-pentanol None of the given 4-methyl-2-pentanol 3-methyl-1-butanolarrow_forward
- Indicate which alcohol in each pair undergoes an elimination reaction more rapidly when heated with H2SO4.arrow_forwardConvert benzene into attached compound. You may also use any inorganic reagents and organic alcohols having four or fewer carbons. One step of the synthesis must use a Grignard reagent.arrow_forwardWhich of the following alcohols will yield a carboxylic acid product upon oxidation with potassium dichromate in an acidic solution (Jones Oxidation)? a. 2-methyl-2-pentanol b. 4-methyl-2-pentanol c. None of the given d. 3-methyl-1-butanolarrow_forward
 EBK A SMALL SCALE APPROACH TO ORGANIC LChemistryISBN:9781305446021Author:LampmanPublisher:CENGAGE LEARNING - CONSIGNMENT
EBK A SMALL SCALE APPROACH TO ORGANIC LChemistryISBN:9781305446021Author:LampmanPublisher:CENGAGE LEARNING - CONSIGNMENT
