
(a)
Interpretation:
It should be determined that the amide used to produce Benzylmethylamine on reaction with
Concept introduction:
LiAH4 (Lithium aluminium hydride):
Lithium aluminium hydride is used as a reducing agent.
Lithium aluminium hydride is reduced the amide as an
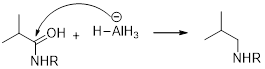
(b)
Interpretation:
It should be determined that the amide used to produce Ethylamine on rection with
Concept introduction:
LiAH4 (Lithium aluminium hydride):
Lithium aluminium hydride is used as a reducing agent.
Lithium aluminium hydride is reduced the amide as an amine which is shown below.
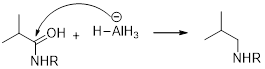
(c)
Interpretation:
It should be determined that the amide used to produce Diethylamine on rection with
Concept introduction:
LiAH4 (Lithium aluminium hydride):
Lithium aluminium hydride is used as a reducing agent.
Lithium aluminium hydride is reduced the amide as an amine which is shown below.
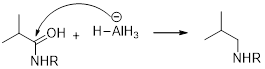
(d)
Interpretation:
It should be determined that the amide used to produce Triethylamine on rection with
Concept introduction:
LiAH4 (Lithium aluminium hydride):
Lithium aluminium hydride is used as a reducing agent.
Lithium aluminium hydride is reduced the amide as an amine which is shown below.
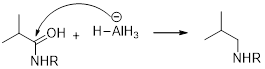
Want to see the full answer?
Check out a sample textbook solution
Chapter 12 Solutions
Essential Organic Chemistry (3rd Edition)
- What are heterocyclic amines? Name two heterocyclic amines associated with structure of DNA and RNA. Explain comprehensively.arrow_forwardWhich statements are TRUE?I. Tertiary amines have lower BP than primary and secondary amines.II. Tertiary amines has no possibility for hydrogen bonding.III. Tertiary amines has a high-molecular mass as hydrogen bonding occursarrow_forwardIs Tripamide soluable in waterarrow_forward
- How to compare an alkylamine (CH3CH2NH2) and an arylamine (C6H5NH2, aniline) ?arrow_forwardExplain Amines ?arrow_forwardN-Methylpyrrolidine has a boiling point of 81 °C, and piperidine has a boiling point of 106 °C. Tetrahydropyran has a boiling point of 88 °C, and cyclopentanone has a boiling point of 141 °C. These two isomershave a boiling point difference of 53 °C. Explain why the two oxygen-containing isomers have a much larger boilingpoint difference than the two amine isomersarrow_forward

 Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning
Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning Chemistry: An Atoms First ApproachChemistryISBN:9781305079243Author:Steven S. Zumdahl, Susan A. ZumdahlPublisher:Cengage Learning
Chemistry: An Atoms First ApproachChemistryISBN:9781305079243Author:Steven S. Zumdahl, Susan A. ZumdahlPublisher:Cengage Learning
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning





