
Concept explainers
(a)
Interpretation:
The Lewis structure and resonating structures (if possible) of given molecule is to be drawn.
Concept introduction:
Lewis structure is a representation of a molecule which shows shared and unshared pair of electrons. It is helpful to determine the shape of a molecule.
(a)
Answer to Problem 36A
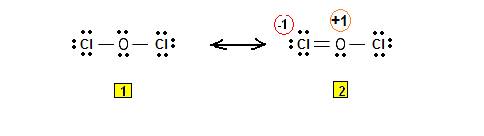
Explanation of Solution
Oxygen has six valence electrons. One electron is shared by each chlorine atom and two electron pairs are left unshared.
Its resonating structure is also possible as given.
(b)
Interpretation:
The Lewis structure and resonating structures (if possible) of given molecule is to be drawn.
Concept introduction:
Lewis structure is a representation of a molecule which shows shared and unshared pair of electrons. It is helpful to determine the shape of a molecule.
(b)
Answer to Problem 36A

Explanation of Solution
Carbon has four valence electrons. Two electrons are shared by each oxygen atom and no electrons are left unshared.
(c)
Interpretation:
The Lewis structure and resonating structures (if possible) of given molecule is to be drawn.
Concept introduction:
Lewis structure is a representation of a molecule which shows shared and unshared pair of electrons. It is helpful to determine the shape of a molecule.
(c)
Answer to Problem 36A
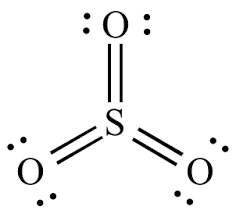
Resonating structures of
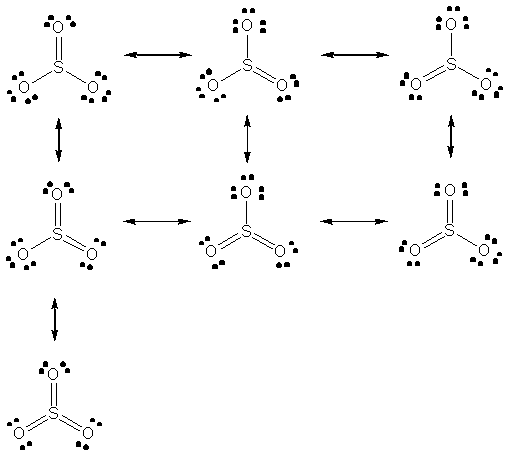
Explanation of Solution
Sulphur has six valence electrons. Each oxygen share two electrons with sulphur and no electrons are left unshared.
Seven resonating structures are possible for
Chapter 12 Solutions
World of Chemistry
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY





