
Concept explainers
(a)
Interpretation:
The dipole moment arrow is to be drawn for the given molecule.
Concept introduction:
Dipole moment arrows are drawn from the atom with less electronegativity to the atom with more electronegativity.
(a)
Answer to Problem 6RQ
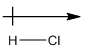
Explanation of Solution
Chlorine is more electronegative than hydrogen. So, the arrow will be from hydrogen to chlorine.
(b)
Interpretation:
The dipole moment arrow is to be drawn for the given molecule.
Concept introduction:
Dipole moment arrows are drawn from the atom with less electronegativity to the atom with more electronegativity.
(b)
Answer to Problem 6RQ
None
Explanation of Solution
The bond is formed between the same atoms. So it is a non-polar compound.
(c)
Interpretation:
The dipole moment arrow is to be drawn for the given molecule.
Concept introduction:
Dipole moment arrows are drawn from the atom with less electronegativity to the atom with more electronegativity.
(c)
Answer to Problem 6RQ
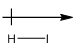
Explanation of Solution
Iodine is more electronegative than hydrogen. So, the arrow will be from hydrogen to iodine.
(d)
Interpretation:
The dipole moment arrow is to be drawn for the given molecule.
Concept introduction:
Dipole moment arrows are drawn from the atom with less electronegativity to the atom with more electronegativity.
(d)
Answer to Problem 6RQ
None
Explanation of Solution
The bond is formed between the same atoms. So it is a non-polar compound.
(e)
Interpretation:
The dipole moment arrow is to be drawn for the given molecule.
Concept introduction:
Dipole moment arrows are drawn from the atom with less electronegativity to the atom with more electronegativity.
(e)
Answer to Problem 6RQ
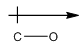
Explanation of Solution
Oxygen is more electronegative than carbon. So, the arrow will be from carbon to oxygen.
Chapter 12 Solutions
World of Chemistry
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY





