
Concept explainers
(a)
Interpretation:
The product of the given reaction is to be predicted.
Concept Introduction:
Addition reaction: A general reaction type in which a substance X-Y adds to the multiple bond of an unsaturated reactant to yield a saturated product that has only single bonds.
Hydrogenation (Addition of
b)
Interpretation:
The products of the reaction to be predicted
Concept Introduction:
Reaction of
Nitration: The substitution of a nitro group for hydrogen on an aromatic ring.
(c)
Interpretation:
The product of the given reaction to be predicted
Concept Introduction:
Addition reaction: A general reaction type in which a substance X-Y adds to the multiple bond of an unsaturated reactant to yield a saturated product that has only single bonds.
Hydration:
When alkene is undergoes hydration with water in the presence of sulfuric acid which yields the alcohol. In this reaction, the water molecule will behave like a hydrogen halide to the alkene which gives the addition product this reaction is known as a hydration reaction.
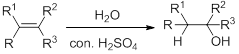
Alkene is reaction with water in the presence of sulfuric acid, first step is proton (
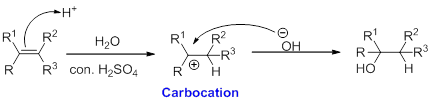
In hydration reaction, sulfuric acid is act as a proton donor, which is the driving force of the reaction. Hydration reaction will not go without acid (sulfuric acid).
Markovnikov’s rule: In the addition of
(d)
Interpretation:
The product of the given reaction to be predicted
Concept Introduction:
Addition reaction: A general reaction type in which a substance X-Y adds to the multiple bond of an unsaturated reactant to yield a saturated product that has only single bonds.
Hydration:
When alkene is undergoes hydration with water in the presence of sulfuric acid which yields the alcohol. In this reaction, the water molecule will behave like a hydrogen halide to the alkene which gives the addition product this reaction is known as a hydration reaction.
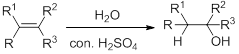
Alkene is reaction with water in the presence of sulfuric acid, first step is proton (

In hydration reaction, sulfuric acid is act as a proton donor, which is the driving force of the reaction. Hydration reaction will not go without acid (sulfuric acid).
Markovnikov’s rule: In the addition of
(e)
Interpretation:
The product of the given reaction to be predicted
Concept Introduction:
Addition reaction: A general reaction type in which a substance X-Y adds to the multiple bond of an unsaturated reactant to yield a saturated product that has only single bonds.
Hydrogenation (Addition of
Want to see the full answer?
Check out a sample textbook solution
Chapter 13 Solutions
EBK FUNDAMENTALS OF GENERAL, ORGANIC, A
- Under standard conditions, will the following reaction proceed spontaneously as written?arrow_forwardIdentify the oxidized reactant, the reduced reactant, the oxidizing agent, and the reducing agent in the following reactions:arrow_forwardDraw out the reactant and product(s) for the following reaction: [0] 16:149 (COMPLETE OXIDATION)arrow_forward
- Propose an appropriate intermediate for the following reactionarrow_forwardDraw the product AND propose a reasonable, detailed stepwise mechanism, using curved arrow notation to show the flow of electrons, for the following reaction.arrow_forwardConsider the reaction below to answer the following question(s): + HBr A B Br с + D Br Enter the appropriate letter in the blank for each the following statements. The kinetically controlled product in this reaction is D B Aarrow_forward
- Why is the term dark reactions misleading?arrow_forwarddiscuss the following statement: “Whether the ΔG for a reaction is larger, smaller, or the same as ΔG° depends on the concentration of the compounds that participate in the reaction.”arrow_forwardGive an example of each Irreversible and Reversible Reactions?arrow_forward
 BiochemistryBiochemistryISBN:9781319114671Author:Lubert Stryer, Jeremy M. Berg, John L. Tymoczko, Gregory J. Gatto Jr.Publisher:W. H. Freeman
BiochemistryBiochemistryISBN:9781319114671Author:Lubert Stryer, Jeremy M. Berg, John L. Tymoczko, Gregory J. Gatto Jr.Publisher:W. H. Freeman Lehninger Principles of BiochemistryBiochemistryISBN:9781464126116Author:David L. Nelson, Michael M. CoxPublisher:W. H. Freeman
Lehninger Principles of BiochemistryBiochemistryISBN:9781464126116Author:David L. Nelson, Michael M. CoxPublisher:W. H. Freeman Fundamentals of Biochemistry: Life at the Molecul...BiochemistryISBN:9781118918401Author:Donald Voet, Judith G. Voet, Charlotte W. PrattPublisher:WILEY
Fundamentals of Biochemistry: Life at the Molecul...BiochemistryISBN:9781118918401Author:Donald Voet, Judith G. Voet, Charlotte W. PrattPublisher:WILEY BiochemistryBiochemistryISBN:9781305961135Author:Mary K. Campbell, Shawn O. Farrell, Owen M. McDougalPublisher:Cengage Learning
BiochemistryBiochemistryISBN:9781305961135Author:Mary K. Campbell, Shawn O. Farrell, Owen M. McDougalPublisher:Cengage Learning BiochemistryBiochemistryISBN:9781305577206Author:Reginald H. Garrett, Charles M. GrishamPublisher:Cengage Learning
BiochemistryBiochemistryISBN:9781305577206Author:Reginald H. Garrett, Charles M. GrishamPublisher:Cengage Learning Fundamentals of General, Organic, and Biological ...BiochemistryISBN:9780134015187Author:John E. McMurry, David S. Ballantine, Carl A. Hoeger, Virginia E. PetersonPublisher:PEARSON
Fundamentals of General, Organic, and Biological ...BiochemistryISBN:9780134015187Author:John E. McMurry, David S. Ballantine, Carl A. Hoeger, Virginia E. PetersonPublisher:PEARSON





