
Organic Chemistry
12th Edition
ISBN: 9781118875766
Author: T. W. Graham Solomons, Craig B. Fryhle, Scott A. Snyder
Publisher: WILEY
expand_more
expand_more
format_list_bulleted
Concept explainers
Question
Chapter 13, Problem 49P
Interpretation Introduction
Interpretation:
The starting material for the given reactions is to be determined.
Concept introduction:
Diels-alder is a type of organic reaction in which a substituted
The general reaction of Diels-alder is as follows:
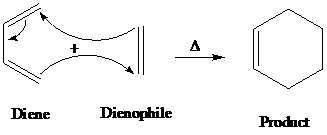
A
The organic reaction in which oxidation of primary alcohol to carboxylic acid and secondary alcohol to ketone takes place is known as Jones oxidation.
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
Please show the electron-flow mechanism of the general synthesis of Benzyl Chloride from Phenol. This involves predicting major and by-products using electronic and structural effects. The arrow push mechanism must be shown.
Make sure you include a detailed explanation of your answer, and provide arrows for mechanisms 'if needed'.
A student was given from the list of the compounds below A, B and D blindly and asked to identify them all. He treated each of them with Brady's reagent (2,4-ditrophenylhydrazine) and isolated a bright yellow compound for one of them, but the other two gave false negatives. The student reasoned that the false negatives may be due to sterics and, on further thinking, it dawned on him that he might be able to rule out one of the false negatives with the haloform test. What compound did he find compatible with the haloform test? That compound did indeed give a false negative in the Brady test. Which of the other two was positive in the Brady test?
A = haloform
B = Brady
A = haloform
D = Brady
B = haloform
A = Brady
B = haloform
D = Brady
D = haloform
A = Brady
D = haloform
B = Brady
Chapter 13 Solutions
Organic Chemistry
Ch. 13 - Prob. 1PPCh. 13 - Prob. 2PPCh. 13 - Prob. 3PPCh. 13 - Practice Problem 13.4 From each set of resonance...Ch. 13 - Practice Problem 13.5 The following enol (an...Ch. 13 - Prob. 6PPCh. 13 - Practice Problem 13.7
Two compounds, A and B, have...Ch. 13 - Prob. 8PPCh. 13 - Prob. 9PPCh. 13 - Prob. 10PP
Ch. 13 - Prob. 11PPCh. 13 - Prob. 12PPCh. 13 - Prob. 13PPCh. 13 - Prob. 14PPCh. 13 - Prob. 15PPCh. 13 - Practice Problem 13.16
Diels–Alder reactions also...Ch. 13 - Prob. 17PPCh. 13 - Prob. 18PCh. 13 - What product would you expect from the following...Ch. 13 - Prob. 20PCh. 13 - Prob. 21PCh. 13 - Provide the reagents necessary for each of the...Ch. 13 - Prob. 23PCh. 13 - Prob. 24PCh. 13 - Prob. 25PCh. 13 - When 1-pentene reacts with N-bromosuccinimide...Ch. 13 - Prob. 27PCh. 13 - Prob. 28PCh. 13 - Prob. 29PCh. 13 - Prob. 30PCh. 13 - 13.31 Provide a mechanism that explains formation...Ch. 13 - 13.32 Provide a mechanism that explains formation...Ch. 13 - Treating either 1-chloro-3-methyl-2-butene or...Ch. 13 - Prob. 34PCh. 13 - Prob. 35PCh. 13 - Although both 1-bromobutane and 4-bromo-1-butene...Ch. 13 - Prob. 37PCh. 13 - Prob. 38PCh. 13 - Prob. 39PCh. 13 - Prob. 40PCh. 13 - Prob. 41PCh. 13 - Prob. 42PCh. 13 - Prob. 43PCh. 13 - 13.44 When furan and maleimide undergo a...Ch. 13 - Two controversial hard insecticides are aldrin and...Ch. 13 - Prob. 46PCh. 13 - Prob. 47PCh. 13 - Prob. 48PCh. 13 - Prob. 49PCh. 13 - Prob. 50PCh. 13 - Explain the product distribution below based on...Ch. 13 - Mixing furan (Problem 13.44) with maleic anhydride...Ch. 13 - Prob. 53PCh. 13 - Prob. 54PCh. 13 - Prob. 1LGPCh. 13 - Prob. 2LGP
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- Working Backwards Parte Deux. a.First, identify which functional groups changed during the reaction. Did stereochemistry change too? b. Based on your answers to A and B, what kind of reaction occurred here, SN1 or SN2? c.Suggest reagents and solvent that could accomplish this reaction.arrow_forwardDraw/outline a reasonable and detailed mechanism for the dehydration of 2-methylcyclohexanol catalyzed by phosphoric acid. Use curved arrows to show the flow of electrons and draw the structures of all intermediates and byproducts formed in the reaction. Predict and rationalize the expected product distribution based on thermodynamic aspectsarrow_forwardUse line structures and write the mechanism for the first and second steps of the hydrobromination reaction of compound C in the presence of HBr to demonstrate that it is a regioselective reaction. Clearly indicate the following: a) the flow electrons in step 1 and in step 2 and b) draw the product D(product 2) resulting from step 2. Include stereo chemistry in your answer.arrow_forward
- (19, 8, 2) Specify a synthetic scheme that would produce the compound shown above in the fewest steps possible. Use one of the starting materials shown together with any of the available reagents. Give the number of the starting material followed by the letters of the reagents in the order of their use, for example: 3be.arrow_forwardThe following equation shows the bromination of compound 1. Propose a structure for product D and a curved arrow mechanism that accounts for its formation featuring the initiation, propagation and termination steps. can you also discuss the stereochemical outcome.arrow_forwardStarting exactly with any acid chloride with exactly with 5 carbon atoms, and using appropriate reagents outline the synthesis of the following molecules: (a) 2,6-dimethyl-4-heptanone (b) 4-propyl-4-octanolarrow_forward
- Draw the structure of the desired dibromide product H. ) Propose a reasonable arrow-pushing mechanism for formation of the byproduct I.Use your mechanism to justify the stereochemistry in molecule Iarrow_forwardStarting with toluene, outline a synthesis of each of the following: (a) o-nitrobenzoic acid (b) m-nitrobenzoic acid (c) 3-chlorobenzoic acid (d) p-bromobenzoic acidarrow_forwardWolff-Kishner reduction of compound W gave compound A. Treatment of A with m-chloroperbenzoic acid gave B which on reduction with LiAH4 gave C. Oxidation of compound C with chromic acid gave D (C9H14O). Suggest the structures for A, B, C, and D.arrow_forward
- Outline a synthesis of the following compounds (A-C) from acetoacetic ester OR malonic ester. Show the starting material and reagents. If the synthesis is not possible, indicate why. A) hept-6-en-2-one B) 2-benzylheptanoic acid C) 3-isobutyl-2-heptanonearrow_forwardPlease show the electron-flow mechanism of the general synthesis of fluorobenzene from aniline, this involves predicting major and by-products using electronic and structural effects. The arrow push mechanism must be shown.arrow_forwardProvide a mechanism which explains the following conversions. Include all intermediates (where appropriate) and watch your arrows and chargesarrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY

Chemistry
Chemistry
ISBN:9781305957404
Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:Cengage Learning

Chemistry
Chemistry
ISBN:9781259911156
Author:Raymond Chang Dr., Jason Overby Professor
Publisher:McGraw-Hill Education

Principles of Instrumental Analysis
Chemistry
ISBN:9781305577213
Author:Douglas A. Skoog, F. James Holler, Stanley R. Crouch
Publisher:Cengage Learning

Organic Chemistry
Chemistry
ISBN:9780078021558
Author:Janice Gorzynski Smith Dr.
Publisher:McGraw-Hill Education

Chemistry: Principles and Reactions
Chemistry
ISBN:9781305079373
Author:William L. Masterton, Cecile N. Hurley
Publisher:Cengage Learning

Elementary Principles of Chemical Processes, Bind...
Chemistry
ISBN:9781118431221
Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. Bullard
Publisher:WILEY