
Concept explainers
Interpretation:
The steps how the two products formed from reaction of methylenecyclohehane with NBS has to be given. Stereoisomers has to be disregarded.
Concept introduction:
Bromination of Allylic Carbons:
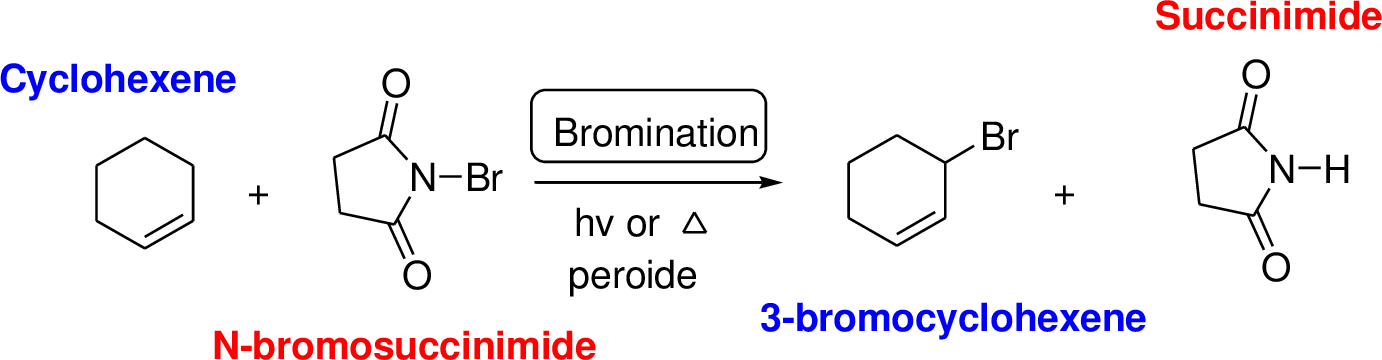
N-bromosuccinimide (NBS) is used for the allylic bromination through radical reaction. Bromination of allylicc carbon requires low concentration of bromine and low concentration of hydrobromic acid. If high concentration of bromine and high concentration of hydrobromic acid which leads to the formation of bromonation in the double bond.
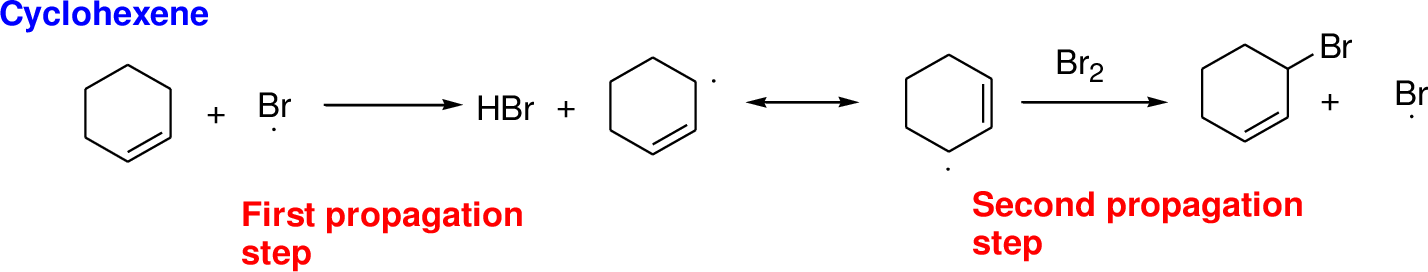
Bromination reaction starts with the homolytic cleavage of N-Br bond in N-bromosuccinimide (NBS) which creates bromine radical to initiate the radical bromination reaction.
NBS bromine radical removes the allylic hydrogen which forms hydrogen bromide and allylic radical in the first propagation step, the allylic radical is stabilized by the double bond in ring. This allylic radical reaction with bromine molecule and forms allylic bromide in the second propagation step which are shown above.
Want to see the full answer?
Check out a sample textbook solution
Chapter 13 Solutions
Organic Chemistry
- Bicyclo-2,5-heptadiene can be prepared in two steps from cyclopentadiene and vinyl chloride. Provide a mechanism for each step.arrow_forwardAccount for the regioselectivity and stereoselectivity observed when 1-methylcyclopentene is treated with reagent. Q) Br2 in H2Oarrow_forwardDraw the products formed when both cis- and trans-but-2-ene are treated with OsO4, followed by hydrolysis with NaHSO3 + H2O. Explain how these reactions illustrate that syn dihydroxylation is stereospecic.arrow_forward
- cis-4-Bromocyclohexanol and trans-4-bromocyclohexanol form the same elimination product but a different substitution product when they reactwith HO-. a. Why do they form the same elimination product?b. Explain, by showing the mechanisms, why different substitution products are obtained.c. How many stereoisomers does each of the elimination and substitution reactions form?arrow_forwardDraw the products formed when both cis- and trans-but-2-ene are treated with OsO4, followed by hydrolysis with NaHSO3 + H2O. Explain how these reactions illustrate that syn dihydroxylation is stereospecific.arrow_forwardHow many stereoisomers are formed from the reaction of 3-methylcyclohexene with NBS?arrow_forward
- Identify two alkenes that react with HBr to form 1-bromo-1-methylcyclohexane without undergoing a carbocation rearrangement.arrow_forwardDraw the products, including their configurations, obtained from the reaction of 1-ethylcyclohexene with the following reagents:a. HBr b. H2, Pd/C c. R2BH/THF, followed by HO– , H2O2, H2O d. Br2/CH2Cl2arrow_forwardConsider a reaction where cis-but-2-ene is treated with OsO4 followed by NaHSO3/H2O. Draw the structure of one product that is formed in the reaction, including correct stereochemistry.arrow_forward

 Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning

